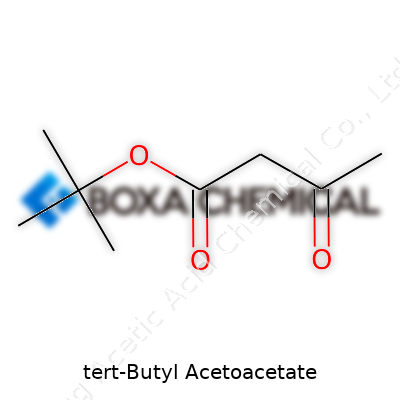
Years ago, chemistry felt like a world of blackboards and glass flasks, with certain reagents quietly changing how people made medicines and coatings. Tert-butyl acetoacetate (TBAA) belongs to this list of behind-the-scenes players. Researchers first started preparing acetoacetic esters over a century ago, where each step drew from German and French chemical traditions. TBAA showed up on the scene as industry began longing for materials with more selective reactivity and stability, driven by pharmaceutical synthesis and paint technology. As interest in environmentally safer processes grew, chemists took a closer look at which protective groups and intermediates made their transformations more practical—and TBAA’s tert-butyl group has kept it relevant ever since.
You rarely see TBAA as a headliner in everyday conversation, but in the lab and plant, it gets steady respect. Its structure combines the acetoacetate backbone known for easy transformations with a bulky tert-butyl ester, a feature prized in modern organic chemistry. This molecule’s flexibility sets it apart: drug discovery teams favor it for making complex intermediates, while coatings manufacturers rely on its selective breakdown behavior. TBAA is not a household name, yet it often sits on the shelves of those working on sophisticated chemical routes or next-generation materials.
This compound pours out as a colorless or slightly yellow liquid, carrying a moderate fruity odor—a far cry from the harshness of earlier reagents used for similar jobs. Its boiling point sits comfortably above what many solvents can handle, which allows for easy manipulation in typical lab conditions. Water doesn’t mix with it, but most common organic solvents do, fitting it right into reaction mixtures for skilled hands to guide transformations. The tert-butyl group protects one end from unwanted reactions, giving chemists a degree of control often required in work that leaves little room for error. Its stability makes storage less of a headache compared to edgier analogues.
Once someone starts sourcing TBAA, purity jumps into focus, especially for critical pharmaceutical projects. The market offers variants commonly marketed above 98 percent purity, but real-world synthesis often requires testing for moisture, acidity, or trace byproducts before use. Sensible manufacturers detail labeling to flag its moderate flammability and the need for ventilation; regulations follow established hazardous material standards and lean on globally harmonized codes. In practice, users respect the same signs, symbols, and documentation that have guided safe chemical handling for decades.
Crafting TBAA in the lab might sound routine now, but it’s a careful dance—chemists react tert-butanol with diketene or acetoacetic acid, all while controlling temperature and side reactions. Reaction efficiency climbed higher over the years with tweaks to catalysts and process design. Lessons from larger-scale synthesis turned up ways to reduce byproducts, streamline post-reaction purification, and improve isolation, which matters most to those who depend on consistent results batch after batch. Continuous flow methods have popped up, taking the edge off the dangers associated with older, batch-based approaches.
You pick up deep respect for TBAA’s versatility after watching chemists lean on it for cross-couplings, condensations, or as a masked acetoacetic acid. The molecule serves as a safer alternative to methyl or ethyl esters when the downstream process calls for extra protection from premature hydrolysis or unwanted rearrangement. Removing the tert-butyl group pinpoints TBAA’s real skill—‘deprotecting’ on command under mild acid conditions, with far less fuss than other protecting groups. Medicinal chemists count on this tool to avoid lost time or tricky cleanups. The range of derivatives, from hydrazones to substituted enolates, keeps it adaptable for the current needs of labs and production plants.
TBAA answers to a handful of aliases in both literature and commerce: tert-butyl 3-oxobutanoate, tert-butyl acetoacetate, and sometimes even 1,3-dioxo-2-butanone, tert-butyl ester. You might spot other translations, but these main handles persist. Clarity in naming clears the way for safer swaps or substitutions across regions and industries, making sure buyers don’t confuse this useful chemical with less suitable cousins.
Lab veterans know to take TBAA’s fire risk seriously. Stable as it seems, its vapors catch easily near flames or sparks, pressing teams to work with it only in properly ventilated spaces and to keep clear of open ignition sources. Direct contact causes irritation, motivating anyone handling it to suit up with gloves, goggles, and lab coats. Storage in dry, cool, well-marked cabinets helps prevent mix-ups and chemical surprises. Spillage brings standard cleanup procedures—adsorbents, good ventilation, and safe disposal of waste. Facilities check for engineering controls to keep exposures within occupational guidelines, as stipulated by recognized safety bodies.
Pharmaceutical research leans heavily on intermediates like TBAA, valuing its controlled reactivity. The compound’s role as a masking group shows up everywhere complex molecules demand careful stepwise assembly—the protection and later removal of sensitive units is a daily fact of modern medicinal chemistry. Agrochemicals and specialty coatings depend on TBAA for similar reasons, especially where final products require consistent performance and stability. Pigments, inks, and even some flavors benefit from its ability to bring in the acetoacetate functionality without harsh processing conditions. Anyone working at these crossroads of innovation approaches TBAA with both utility and respect.
In research groups and startup labs, TBAA invites curiosity for both classic and new chemistry. Experts probe its use in asymmetric synthesis, while greener process advocates look at swapping out less forgiving acylation sources. Modifying the tert-butyl group, or choosing selective catalysts for milder conditions, sparks a lot of published work—and every tweak promises safer, leaner workflows. Computational chemists explore reactivity patterns to design routes that use less energy, which matches demands from companies trying to cut emissions and curb waste. Each wave of discovery breathes fresh purpose into simple molecules like TBAA, showing that innovation can surface from the chemistry bench as often as from star R&D labs.
Evaluating TBAA’s health impacts teaches humility. Acute toxicity studies show moderate risk if swallowed or if vapors get inhaled in poorly controlled work areas. Reports document irritation to eyes, throat, and sometimes skin, which supports the use of commonsense barriers when loading reactors or transferring to bottles. Long-term exposure data remains far from comprehensive, but results so far do not point to carcinogenic effects at exposure levels found in industrial or lab environments. Still, users talk openly with safety consultants, collecting monitoring data and adjusting practices to close any gaps. Animal studies—where available—reinforce proper handling advice, and regulators keep a close watch for any emerging risks as use grows.
Sitting on the edge of what’s possible, chemists and manufacturers look at TBAA through the lens of sustainable process design and next-generation materials. Pressure to replace hazardous reagents with safer, more selective intermediates lands the tert-butyl ester family in a strong position for growth. Green chemistry pushes the push for recyclable protecting groups, reactions that create less waste, and steps that lower energy use—sparking creative attempts to reinvent TBAA or its cousins for upcoming manufacturing challenges. Medicine keeps raising the bar for reliable, high-purity intermediates, meaning steady demand. With the global shift toward cleaner, more responsible production, TBAA and similar compounds will likely stick around, earning their place as essential tools in the pursuit of safer, smarter chemistry.
Walk into any organic chemistry lab, and you’ll spot tert-Butyl Acetoacetate sitting among the glassware and solvents. This isn’t just another bottle on a shelf, but a backbone for transformation in the hands of chemists who work to build complex molecules. I remember the first time I worked with it during my undergrad days, trying to put together a coumarin molecule for a fluorescence experiment. The process felt almost magical—the way a simple ester could open doors to something much bigger.
If you look at the pharmaceutical industry, you’ll see tert-Butyl Acetoacetate at the starting line for many syntheses. Chemists value it for making heterocycles, the kind you find at the center of medicines that treat heart conditions, infections, and sadly, cancer too. Its role feels almost unsung—without this ester, a chunk of those drug candidates just don’t come together as smoothly.
The colorants industry owes it plenty, as well. I’ve seen dye chemists use tert-Butyl Acetoacetate to build coumarins and similar compounds that light up highlighters, inks, or even certain textiles. The ease with which this ester lets you attach different functional groups matters a lot; it saves steps, reducing waste and cost, and that efficiency means both established and smaller labs can keep exploring new applications.
Having worked with similar chemicals, what stands out about tert-Butyl Acetoacetate is its stability. It doesn’t give you the trouble that some beta-keto esters will—if you’ve ever tried to store ethyl acetoacetate for long stretches, you know the frustration. In the hands of a chemist, this means you can run reactions at higher temperatures or in open air more confidently, cutting the risk of expensive mistakes.
It also gives you predictable, clean products especially during alkylation or condensation reactions. That’s huge in the lab—less purification means you get to the next step faster, and you waste less material. Chemists always look for shortcuts that keep their experiments smooth, and tert-Butyl Acetoacetate delivers them.
Handling tert-Butyl Acetoacetate does raise environmental and safety questions. Like most organic solvents and esters, there’s the risk of exposure and issues if large quantities aren’t disposed of correctly. I’ve seen labs reduce risk by shifting to safer work-up procedures, such as using closed systems to limit vapors, or waste treatment setups that neutralize remnants before they reach waterways.
On the green chemistry front, there’s space to improve. Companies could develop biosynthetic pathways for making the precursor acids, cutting down on fossil input. Researchers talk a lot about greener solvents and reusable catalysts, and integrating these with tert-Butyl Acetoacetate chemistry seems like a realistic path. Instead of relying only on what’s always worked, it makes sense to push toward setups that are both cost-effective and environmentally sound.
For industries where flexibility and high yield matter, tert-Butyl Acetoacetate fits the bill. My own hands-on memories and the stories from colleagues show the value this molecule brings to the table. With tighter safety practices and a push for greener synthesis, its role in forming medical, agricultural, and dye compounds isn’t just safe—it’s sustainable.
Back in my graduate days, nothing tripped up a new chemist faster than tripping over the wrong chemical. Ask for “tert-Butyl Acetoacetate,” and the formula should be as familiar as your home address. It’s written as C8H14O3. The CAS number that ties up paperwork, orders, patents, and lab notes is 1694-31-1. This combo isn’t just trivia. Every single researcher, quality control staffer, and logistics handler relies on these details to keep the gears turning and to stop small mistakes before they become disasters.
Plenty of folks see chemistry as a soup of similar-sounding stuff—one “tert-butyl” or “ethyl” away from confusion. In practice, dropping a digit or a letter means buying an entirely different chemical or, worse, causing a bad reaction. I once watched a team rush an entire pilot batch after someone ordered ethyl acetoacetate instead of tert-butyl acetoacetate. Similar names, different properties. This isn’t just annoying. Time, money, and trust all end up at stake.
Small molecules, those like tert-Butyl Acetoacetate, carve out space in pharmaceuticals and coatings, and they spark ideas in university labs just as often as on commercial floors. The chemical formula C8H14O3 isn’t just a badge; it’s a molecular handshake. As for the CAS number 1694-31-1, it’s a ticket that travels across customs agents and safety data sheets all across the world, making sure what you get matches what you intend to use.
Modern regulations demand tight oversight. One misstep can trigger recalls, fines, or worse, hurt someone. In my work on small-scale synthesis, we checked every identifier twice, cross-referencing supplier and regulatory documents. Digital product catalogs and chemical registries take these numbers, run them through software, and merge that data with shipping documents, customs filings, and patent applications. If these don’t line up, delays pile up. Missed deadlines trigger a mess across production and research teams.
Misidentification also slows down progress. Research that builds on the wrong chemical doesn’t just cost money. It wastes months or years. One bad bottle in a crowded storeroom can create noise through dozens of experiments, pushing researchers to chase ghosts that never existed. With tert-Butyl Acetoacetate, purity and identity are closely watched not just for process reliability but to stop cross-contamination, especially in sensitive pharmaceutical production.
Trust has roots in transparency. Coders can tell you: garbage in, garbage out. The same applies here. Every accurate formula or number builds confidence across research, production, and commercial teams. When someone reads “tert-Butyl Acetoacetate,” paired with C8H14O3 and CAS 1694-31-1, they know what’s showing up in the drum or vial. Labs and factories get safer because people spot errors early.
Keeping chemical data accurate and accessible involves more than training or the latest AI platform. It relies on grit and culture. Double-checking is a habit, not a policy. Encouraging open questions means someone catches a typo before it’s too late. Investing in people and systems to keep this process honest pays for itself every time a bad batch gets stopped at the door. Reliable science, meaningful safety, and less waste all tie back to facts that never change, no matter the job title or project scope.
A lot of folks walk into a chemical storeroom or a lab and see a bottle labeled tert-Butyl Acetoacetate, shrug, and stash it on the shelf with everything else. This kind of indifference can create bigger problems than people expect. I’ve learned over the years in labs and chemical stockrooms that a little bit of care can save a mountain of regret. tert-Butyl Acetoacetate isn't some run-of-the-mill solvent you can ignore. It brings hazards to the workspace, and those refusing to pay attention usually end up on the wrong side of a chemical safety incident.
tert-Butyl Acetoacetate can catch fire. It’s not as explosive as some chemicals, but the vapors can ignite if there's a spark or open flame nearby. Some chemists think just screwing the cap on tight makes things safe enough, but that’s ignoring how unpredictable chemical vapors prove to be. Storing this ester in a cool, well-ventilated place isn't just a box to check — it’s common sense learned from one too many “close calls.”
Spills make another story. During my years as a lab tech, I saw more than one instance where a casual knock or slip left the floor slick with material. These moments teach fast: tert-Butyl Acetoacetate stings the skin and eyes. Vapors can irritate throats. Gloves and goggles always sit on my workbench, not by habit but by necessity. Chemical burns and respiratory discomfort leave lasting memories, and the best way to avoid them is to never underestimate the risk — even on a routine day.
The chemical itself doesn’t like water or strong bases. Any moisture mixing in can set off unwanted reactions. If you've spent time in chemical storage, you can see which containers are left open without a second thought. I’ve found the opposite approach works better: close containers right away, store away from moisture, and separate from substances like strong acids or bases. This isn’t just a theoretical precaution. In real-life cases, cross-contamination has ruined expensive batches and set off chain reactions that brought entire lab operations to a halt.
The most respected lab managers I’ve worked with post clear signage and keep tert-Butyl Acetoacetate away from heat and ignition sources. They teach everyone—whether it’s a new grad student or a seasoned tech—to check storage shelves and double-check labels before stacking bottles together. I picked up the habit of dating every container and rotating old stock forward, which cuts the risk of forgotten, degrading batches left to ruin.
Training needs to be more than a video and a quick quiz. Hands-on demonstrations, spill drills, and equipment checks drive home what’s at stake. Providing good, well-maintained fume hoods and keeping the ventilation system running smooth may not look glamorous, but it makes a difference in air quality. The shared culture in a lab or warehouse should value respect for these chemicals, built off experience, peer reminders, and stories of what happens when shortcuts become the norm.
Tough regulations get written after accidents pile up. Smart teams get ahead of them. Safety data sheets help shape routines, but vigilance and teamwork matter more. Stock managers I know run audits that catch expired or leaking containers before accidents occur. They set accountability for every step, from receiving inventory to the final disposal, so nobody claims ignorance after something goes wrong.
In the end, handling tert-Butyl Acetoacetate with respect keeps people out of harm's way. It doesn't take a stack of regulations or a list of warnings—just an honest recognition of real risk, some solid habits, and a team that cares enough to remind each other what’s at stake.
Most folks won’t stumble across tert-Butyl Acetoacetate in their daily lives, unless chemistry is part of the job or passion. Used in labs and manufacturing, this colorless liquid gets poured into a variety of roles, from the ingredients in paints to pharma starting materials. Like plenty of specialty chemicals, its smooth journey from bottle to finished good can snarl into danger if mishandled or misunderstood.
I’ve spent time in spaces where the air tingles with unfamiliar scents and beakers bubble on a warm plate. Anyone who’s handled reagents knows that respect for chemicals builds one’s safety instincts. Tert-Butyl Acetoacetate isn’t known for explosive fame or catastrophic cases, but treating it with a shrug could turn a workday into a hospital visit. According to GHS classification, this compound carries irritant warnings. Its vapor stings the nose and eyes, and exposure to skin leaves a raw, burning patch if you’re not careful. Accidental splashes rarely end in disaster, yet repeated skin contact or careless inhalation sets the stage for longer-term discomfort.
Calluses from attitude, not gloves, mark someone who brushes off the risk here. The issue doesn’t revolve around shocking poisonings. Toxicology records show tert-Butyl Acetoacetate lands in the “moderately toxic” territory when taken in by mouth, and serious harm needs large quantities. The real bite comes from the ordinary scenario — a busy chemist distracted by emails, goggles left at the desk, or a spill ignored because it doesn’t seem like much.
Many chemicals make headlines only after a catastrophe. Tert-Butyl Acetoacetate rarely plays the villain in news reports. Still, healthy skepticism beats overconfidence every time. I can remember colleagues who went home with headaches or rashes, downplaying the culprit because "it wasn’t the dangerous stuff." Yet even mid-level hazards, if handled with routine carelessness, can turn a safe workplace sour.
Proper storage and clear labeling start the process. Good ventilation means fumes don’t linger. Training everyone the same way — lab managers and rookies — cuts confusion and keeps old hands honest. Protective gloves, goggles, and fume hoods do more than collect dust. These steps prove more useful than any list of forbidden drinks or “No Smoking” stickers. I’ve seen tough lab techs ignore advice, then blame the air or the detergent for symptoms they could’ve dodged with better habits.
Solid information matters. CDC and OSHA remind users not to eat, drink, or store food near open chemicals. Washing hands, no matter how quick the lunch break, keeps even small-scale exposure from creeping up. Industry knows repeated exposure causes cumulative risk, so making sure everyone on a team trusts and understands safety routines means fewer surprises over time.
Chemical makers and distributors should share up-to-date safety data and offer real-world training instead of thick manuals gathering dust. Online resources keep evolving, but nothing replaces a culture where questions about risk get real answers instead of shrugs. Proper disposal and spill management deserve equal attention, since dumping leftovers down the wrong drain can pile on trouble for a community, not just one lab.
Decades in research taught me that chemicals with an “average” danger rating can lull folks into carelessness. Tert-Butyl Acetoacetate may not be the top villain on toxicology charts, but a measured blend of respect, information, and constant vigilance goes a long way in making sure it never earns the spotlight in an incident report.
Recognizing quality in chemicals like tert-butyl acetoacetate isn’t just for the folks in lab coats. Plenty of students, researchers, and small manufacturers depend on a steady supply of this compound, whether they’re pushing the boundaries of new pharmaceuticals or dialing in a better process for specialty chemicals. What matters on the ground often gets boiled down to two things: purity and physical characteristics. Getting either of these wrong can derail months of hard work.
Most people buying tert-butyl acetoacetate expect upwards of 98% purity. Sometimes you’ll see grades reaching 99% or even higher. These purity levels are not just for peace of mind. Chemistry reacts to the smallest of contaminants. A trace of the wrong byproduct can mean the difference between a clean reaction and hours of troubleshooting.
Labs usually confirm advertised purity using gas chromatography and sometimes NMR when extra proof is needed. In big academic labs, it’s not uncommon to double-check what shows up in the bottle. Real-world experience teaches that sometimes batches differ, especially if storage conditions shift or suppliers cut corners. Purity also decides how much effort gets sunk into downstream purification processes. The higher the starting quality, the less waste and headache later on.
Tert-butyl acetoacetate usually pours out clear and colorless. People used to handling solvents and esters can spot an off-yellow color from across the bench. Small color changes often whisper about aging, contamination, or poor sealing of storage containers. It doesn’t take an advanced chemist to question a murky sample.
This liquid feels slick between the fingers, but most users learn to keep it away from direct contact. Simple checks—say, holding the bottle up to a light—reveal haze or suspended particles that might hint at problems. It sounds basic, but these first impressions often prevent wasted time later. If a clear product shows signs of separation, it’s an instant red flag.
Manufacturing flaws, improper storage, and heat exposure tend to cause trouble. Over the years, I’ve opened supposedly fresh bottles that smelled off or had suspicious yellow tints. Air and light accelerate breakdown in many organic esters, and tert-butyl acetoacetate is no exception. Mistakes in handling can mean the sample is already on a path to decomposition before it reaches the buyer.
Regulatory bodies encourage suppliers to supply certificates of analysis, but there’s no substitute for a quick reality check in your own lab. I recall cases in grad school where a “fresh” batch set off a long troubleshooting hunt, only to realize the culprit was a tainted reagent.
If purity matters, working with established suppliers pays off. Companies with strong reputations tend to invest in better storage, cleaner manufacturing, and truthful labeling. Rotating stock regularly can also limit problems tied to age. For anyone running reactions that count on tert-butyl acetoacetate, making a habit of checking appearance and running quick purity tests offers cheap protection against ruined experiments.
Simple routines and skepticism toward anything odd in the bottle preserve time, money, and effort. Chemistry, after all, punishes shortcuts but rewards attention to detail and a willingness to trust your own senses.