Chemistry fills its timeline with compounds often overshadowed by their more celebrated cousins. N-Methylacetoacetamide, for instance, doesn’t commonly star in popular science tales, but its development traces back to the era where chemists began to explore the power of simple alkylations and amide formations. Early syntheses drew on foundational amide chemistry practiced since the 19th century. Scientists found ways to craft N-Methylacetoacetamide by reacting methylamine with acetoacetic esters, noting the compound’s distinctive properties and usefulness as more than a simple reagent. Over time, research papers highlighted its promise in facilitating synthesis routes and catalyzing small but important changes in various chemical processes.
N-Methylacetoacetamide might sound niche, but its structure—melding an acetoacetamide backbone with a methyl group attached to the nitrogen—gives it features that catch the eye of medicinal chemists and industrial laboratories. Organic chemists bank on its stability, the selectivity it offers in reactions, and the possibilities for modification both at the carbonyl and amide nitrogens. Whether looking to build up more complex molecules or tweak reactivity, this compound steps up as a reliable tool. Its clear, colorless liquid state makes handling it straightforward, which regularly wins points in labs that value simplicity and predictability over unpredictably hazardous agents.
The physical properties of N-Methylacetoacetamide deserve attention because they influence not just how the substance behaves on a bench top but also how it persists or transforms in more dynamic chemical environments. Volatility matches closely with other acetoacetamide derivatives, making airtight storage essential. The boiling point, coupled with low to moderate water solubility, puts it in a category handled with both ease and caution. In my experience, this means you don’t worry about dehydration, but you also keep an eye out when heating or concentrating the material. The methyl group on nitrogen shifts basicity and reactivity just enough to open unexpected doors for synthetic transformations.
Anyone who has handled N-Methylacetoacetamide knows the importance of clarity in labeling. Whether you’re working in a research lab or an industrial plant, understanding concentration, batch purity, and exact storage guidelines is more than a regulatory box to check; it determines your outcome’s success. Careless labeling leads to accidental cross-reactions, so explicit technical details (like melting point, purity, and form) become a silent kind of insurance policy. The wrong solvent, missed contaminant, or slight impurity gets magnified down the line—so labeling and specification sheets act as a critical link in safe and successful application.
Laboratory-scale syntheses tend to stick with tried-and-true methods. Methylamine’s reaction with acetoacetic ester under controlled conditions stands out for its reliability and avoidable side products. Chemists carefully monitor temperature and time because the yield drops sharply as conditions get too hot or go on too long. Scaling up introduces some obstacles—controlled addition, temperature management with larger volumes, and dealing with byproducts on a bigger scale all require problem solving. Improvements in purification—crystallization, careful distillation, or even chromatographic methods—mean the end product today tends to deliver high reliability, but cutting corners leads to frustration later in research or manufacturing.
N-Methylacetoacetamide doesn’t just sit passively in flasks. The molecule invites a range of transformations. The methylene group sandwiched between carbonyls often gets exploited for alkylation, condensation, or cyclization. The nitrogen’s methyl group limits hydrogen bonding, steering the molecule into specific paths in multi-step syntheses. Personally, I’ve found success using it to build β-keto amide frameworks that would otherwise spit out a mess of tars and side products. Its behavior in the presence of electrophiles also delivers distinct advantages—cleaner outcomes where less selective amides turn ugly—so a synthetic chemist finds in it a trusty ally for pursuit of high yield and clean reaction profiles.
You might spot N-Methylacetoacetamide in the literature under names like N-Methyl-3-oxobutanamide or even 3-Oxobutanamide, N-methyl-. Sometimes the shorthand “NMAA” shows up too. Consistency in naming saves more than confusion. In the world of chemical procurement and regulatory paperwork, missing or misusing synonyms can trigger costly mistakes. Scientists swapping samples or data need to agree on terms to avoid missteps, especially when closely related compounds offer subtle but important differences. Regulatory listings, patents, and journals all lean on standardized naming to sidestep ambiguity and streamline discovery and reporting.
Safe handling of N-Methylacetoacetamide comes down to common sense and learned habits. This compound doesn’t erupt in flames or fume violently, but skin exposure, inhalation, or accidental ingestion can deliver unwanted surprises. Chemical users work with gloves, splash protection, and proper ventilation for peace of mind and to avoid lost time to accidents or mishaps. Regulatory standards—local and global—define thresholds, exposure limits, and waste disposal protocols. Companies and research labs commit to ongoing training, audits, and improvements to ensure even seasoned chemists don’t fall into complacency. Good practice saves time, money, and health; safety lapses cost far more than documentation ever will.
Traditional laboratories reach for N-Methylacetoacetamide for its ease in enolate chemistry and as a precursor in small molecule development. Drug discovery teams explore its structure for drug-like fragments, while polymer research explores its compatibility in assembling more complex frameworks. Within agrochemistry, the molecule sometimes takes part in synthesizing active intermediates for herbicides or pesticides. As one delves into organic electronics and the development of new materials, researchers study its reactivity profile to control molecular architecture. My discussions with colleagues point to a steady revival in the interest around such amides, due partly to their balance of stability and selective reactivity—a valuable combination for driving new innovation in crowded fields.
Every time I investigate recent literature, N-Methylacetoacetamide sits somewhere in the background, waiting for its breakout role in new synthesis strategies. Research teams continue to publish on how slight tweaks to its core structure unlock access to previously hard-to-reach molecules. Modern analytical tools like NMR, mass spectrometry, and advanced crystallography enable more thorough analysis, revealing subtleties behind its reactivity and offering new theories about its role in catalysis. R&D efforts aren’t just academic. Companies probing new pharmaceuticals, specialty polymers, or agricultural chemicals invest in understanding how and why this simple amide behaves so effectively under diverse reaction setups. Its presence in emerging patents hints at a quiet but ongoing resurgence in industrial circles, shaping the future quietly but steadily.
No discussion around chemical use today skips over toxicity evaluation and human health implications. Early reports on N-Methylacetoacetamide focused more on its utility than its dangers. Over the past decade, as toxicology testing advanced, more detailed studies began to fill gaps, drawing on both rodent models and cell line assays to measure acute and chronic toxicity. Some findings suggest relatively low hazard compared to more reactive acetamides, but responsible handling remains the baseline. Accidental exposure delivers mild to moderate irritant effects through skin contact or inhalation; long-term effects currently receive attention but so far suggest limited systemic impact. Integration with regulatory compliance—whether OSHA, REACH, or other standards—calls for transparency, ongoing study, and clearly disseminated safety sheets so all users share a common understanding of risks and mitigations.
By watching how the needs of chemical industries and research communities shift, you can sense that N-Methylacetoacetamide’s fortunes remain tied to how much appetite there is for practical, modifiable starting materials. As more synthetic challenges demand selective, versatile amides that don’t cost a fortune or risk regulatory headaches, its value continues to rise. Advances in green chemistry and environmentally conscious processes want reagents with low toxicity, easy derivatization, and minimal waste—a checklist N-Methylacetoacetamide can often meet. Looking into the future, ongoing work around drug intermediates, new polymers, or smart agricultural chemicals will keep this compound in active play. Years of incremental improvements in both synthesis and handling slowly but surely expand its role far outside its original, narrowly defined use case.
I’ve always liked digging under the surface of the chemicals that do the quiet work in labs, far from the limelight. N-Methylacetoacetamide is one of those unassuming molecules. It usually gets tucked away in textbooks under synthetic intermediates, but its role in research and industry tells its own story.
N-Methylacetoacetamide often steps into the shoes of a go-between in organic chemistry. Its molecular structure allows it to help build more complicated compounds. You probably won’t find it on a drugstore shelf, but it’s spent years behind the scenes helping create pharmaceuticals, agricultural products, and colorants. Most chemists see it as a handy building block because the carbonyl and amide groups give a lot of flexibility for reactions.
During my time in university labs, I watched researchers reach for this compound when piecing together new molecules in multi-step syntheses. In drug research, for example, variations of acetoacetamides wind up as links in the chain that leads to finished medicines. The modesty of the molecule belies the kind of innovation it has enabled in drug discovery, especially in routes that need a reliable, somewhat reactive intermediate that won’t cause unnecessary side reactions.
Out in industry, chemists choose N-Methylacetoacetamide for efficiency in producing dyes and pigments. Its chemical nature allows for substitutions that lead to variations in color and performance. The safety profile also beats more hazardous amines, reducing exposure risk, which means a friendlier lab environment and fewer headaches over waste disposal.
Producing specialty chemicals sometimes boils down to picking compounds that won’t break the bank or hold up the timeline. N-Methylacetoacetamide gets high marks for being relatively simple to handle and incorporating well with established manufacturing pipelines. While large chemical firms probably don’t showcase this ingredient in glossy brochures, they rely on it for consistency and predictability. That reliability cuts down on delays in delivering everything from textile dyes to pesticide precursors.
Students and faculty still use it as a template for teaching chemical transformations. The molecule gives everyone a straightforward way to demonstrate nucleophilic substitution or enamine chemistry. I’ve seen undergraduates use it for classic reactions that remain the gold standard for grasping basic concepts before moving on to more complex procedures.
While N-Methylacetoacetamide does its job well, there’s always room for improvement in how chemicals like this one are sourced, handled, and recycled. As demand for greener chemistry grows, producers face pressure to cut back on solvents and optimize yields. Sometimes that means tweaking the synthetic pathway or looking into catalysts that get more mileage out of each batch produced.
Another hurdle revolves around waste. Many academic labs wind up treating leftover or degraded material as hazardous waste without much thought for reclamation. Companies might explore new protocols to recover and reuse waste streams, both cutting costs and shrinking the environmental footprint. Teaching the next generation of chemists how to value every gram also makes a difference.
N-Methylacetoacetamide isn’t one of those splashy headline chemicals, but the things it helps to build ripple out across industries and research. Watching its impact reminds me that science often moves forward on a foundation of small, reliable helpers—shaping the materials and medicines people use every day.
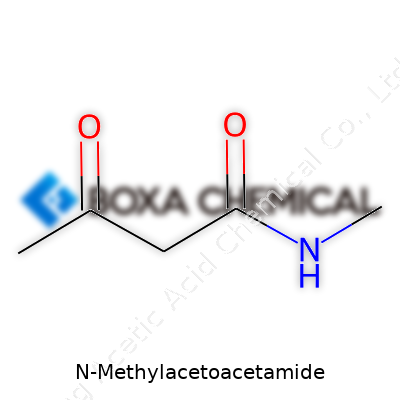
Every time someone handles a chemical in a lab or on a factory floor, they count on a clear understanding of what that material actually is. Science moves fast, and so does industry, but knowing the basic identity of a chemical like N-methylacetoacetamide lays the groundwork for everything that follows. Whether someone plans to synthesize it, store it safely, or use it as a building block in drug development, the formula C5H9NO2 spells out what’s really in the bottle. Even a small error—one atom off—can shift the properties, mix up results, or cause safety issues. Relying on clear facts, not guesses, turns chemical names from jargon into real, understandable substances.
Chemistry might seem far removed from daily concerns, but most innovations start with compounds you’d probably never encounter directly. Take N-methylacetoacetamide as an example. It sits as a quiet contributor in pharmaceutical research and specialty chemical syntheses—fields that eventually touch everyone through medical treatments or advanced materials. Its chemical structure comes down to the combination of a methyl group, an acetoacetamide backbone, nitrogen, and oxygen atoms in specific spots. That’s succinctly captured by C5H9NO2. For scientists and workers, it’s not just a sequence of letters and numbers—it’s a blueprint they follow in the lab.
Looking back at my time in research labs, formulas like this weren’t just abstract knowledge. Each symbol stood for safety checks and the potential for discovery. Long days and nights were organized around making sure each batch matched its intended chemistry. Someone might ask, “Why double check the formula?” After seeing projects stumble because of cross-contamination or labeling mix-ups, the value of clear chemical identities stuck with me. C5H9NO2 becomes more than a formula; it’s trust, built up through shared knowledge.
Missteps happen when formulas get overlooked or glossed over. In educational settings, skipping the step of grounding discussions in actual molecular structures leads students off course. In business, lack of attention to concrete details risks confusing buyers and sellers or, even more critically, undermining safe handling. There’s an urgent case for making chemistry accessible—breaking it down, keeping communication honest, and never assuming everyone knows the facts without discussion.
Start with verified sources. Reputable textbooks, peer-reviewed journals, and recognized chemical databases serve as touchstones. Colleagues in the lab will often remind each other to double check data and formulas—simple habits that prevent bigger headaches down the road. Keeping up with advances, checking specifications against fresh research, and pushing for clear sharing of data in every discussion helps keep errors in check. Passing these good habits on, not just to new scientists but to anyone handling chemicals, strengthens safety and reliability everywhere these compounds go.
You don’t usually hear about compounds like N-Methylacetoacetamide on the evening news. It’s a mouthful, tucked away in research labs and manufacturing plants. Still, questions about its safety don’t stay locked behind laboratory doors. Anyone who has ever handled chemicals in a classroom or workplace knows a subtle anxiety about unknown hazards. That caution keeps a lot of people safe.
N-Methylacetoacetamide comes from a class of organic compounds known for their range of applications. But the biggest question outside the world of chemistry is also the simplest—will this stuff harm me? Experience tells me to look at two things: what the science says and how people actually use the chemical.
To figure out toxicity, I usually start with data from recognized bodies like the National Institutes of Health or the European Chemicals Agency. For N-Methylacetoacetamide, that search leaves a gap. There isn’t much public information on health effects, probably because not many people work directly with it. That doesn’t mean it’s safe. It just hasn’t drawn enough attention—yet.
If you grew up around any industrial town, you learn pretty fast why treating chemicals with respect matters. Even compounds that look tame on paper can pack a punch under the right (or wrong) conditions. For example, simple solvents or additives—once overlooked—have landed on restricted lists only after years of exposure led to health problems. The issue: harm often shows up long after the first spill or careless glove-off moment.
The name N-Methylacetoacetamide signals an amide structure, which tells chemists a little about how it might behave. Amides sometimes irritate the nose or skin. Sometimes they don’t even make you sneeze. But chemical relatives aren’t identical twins. Each structure tweaks toxicity. N-Methylacetoacetamide hasn’t earned a notorious reputation yet, but that could change if industrial use takes off or workers start noticing symptoms.
Walking through any chemical facility, I always notice the big drums with clear labels, gloves clutched by workers, and rinse stations sitting near the exits. These aren’t overkill—they’re a result of hard lessons. People grew sick from things nobody flagged as dangerous. Until more research gives this compound a clean bill of health, treating it with caution makes sense.
Regulations often lag behind discovery. Just because a chemical doesn’t carry a hazard symbol, doesn’t take it off the hook. More chemicals have joined lists of suspected toxins in the past decade, driven by new data and people connecting the dots between symptoms and invisible exposures.
It’s not enough to wait for warning labels. If workers or students find themselves using N-Methylacetoacetamide, I’d want solid training, good ventilation, and protective gear. Management should press for new research and toxicology tests, not just to check boxes, but to stay ahead of problems. Transparency should run through the entire supply chain—nobody benefits from secrets or silence.
Science only moves as fast as the investment behind it. There’s value in giving researchers the money and resources to nail down answers on chemicals like this. Companies and regulators can save money and lives by identifying risk early, not patching it up after the fact.
Nobody wants to see N-Methylacetoacetamide make headlines for the wrong reasons. Until data say otherwise, the responsible move is preparation—not panic. That’s the way progress works in chemistry, and it’s how real trust gets built between industry, workers, and the communities around them.
Anyone who’s worked with chemicals knows the challenges stretch beyond the laboratory bench or warehouse shelves. Talk to any chemist or supply manager, and the conversation usually turns practical: how do we keep this stuff safe until we need it? N-Methylacetoacetamide isn’t a household name, but it deserves careful handling. Missteps in storage—sometimes small lapses—can mean ruined stocks, wasted money, or hazards that weren’t on anyone’s radar.
N-Methylacetoacetamide brings a specific set of risks. It’s an organic compound, and just like many similar chemicals, it doesn’t take kindly to damp corners, sunlight, or fluctuating room temperatures. Stories float around chemical supply rooms about solvents gone bad after being stuck by a leaky window or stacked too close to a steam pipe. Spoilage leads to unnecessary disposal costs and major delays on time-sensitive projects.
From my own days organizing aging chemical closets, I’ve seen the result of casual handling: sticky bottles, illegible labels, containers that crystallized before their time. Mishaps like these wasted hours and forced unnecessary purchases—costly for any team with a budget.
The best chemical safety comes from habits, not from reading a manual once and moving on. With N-Methylacetoacetamide, the advice stays simple:
Regulations from OSHA and comparable bodies ask for robust chemical inventory and hazard training. Authorities push for these not out of bureaucracy, but because real incidents—like accidental mixing or warehouse fires—start from overlooked storage details. Following up-to-date Material Safety Data Sheets doesn’t just tick a box; it prevents emergencies.
A few simple routines stop small mistakes from piling up. Assign responsibility for chemical inventories, so the buck never disappears. Set up storage guidelines that match the local environment, especially for heat or moisture. Equip storerooms with easy-to-read signage, and train every hand that ever reaches for a bottle.
N-Methylacetoacetamide isn’t special because of its name. It’s another reminder that caring for chemicals means caring for co-workers, budgets, and long-term projects. Proper storage proves itself not in the smooth days, but in the emergencies you never have because you did the basics right.
N-Methylacetoacetamide occupies a spot on lab shelves for good reason, though most folks outside a chemistry department rarely cross paths with it. In practice, it shows up more often in places that shape lives quietly but persistently—factories working on specialty chemicals, pharmaceutical labs searching for new treatments, coatings you see on products in stores. Having spent years in both research and industry environments, I’ve seen its uses reach far beyond one narrow role, despite its low profile.
Chemists often turn to N-Methylacetoacetamide for synthesizing pharmaceutical intermediates. One example jumps to mind from work on active pharmaceutical ingredients: the compound can serve as a stepping stone in constructing more complex molecular structures. Some companies favor it during the process of introducing methylated groups, which helps improve a drug’s stability or its bioavailability in the body. Regulatory data suggests patented medicines often include derivatives built around this chemical scaffold. Because purity matters in medicine, N-Methylacetoacetamide finds attention due to its predictable reactivity and the ability to yield high-quality, consistent results batch after batch.
Digging into the coatings industry, there’s a drive to make products tougher, longer-lasting, or more resistant to weather and chemicals. The chemical’s structure fits these demands, helping create resins or adhesion promoters. Industrial users—for instance, paint makers refining formulas to stand up to heat or industrial solvents—have relied on acetoacetamide derivatives for decades. These tweaks improve the look and resilience of paints seen on everything from bridges to cars, something I realized during a stint consulting for a coatings team aiming to beat corrosion without nasty byproducts.
In university research, N-Methylacetoacetamide finds use as a building block for new molecules. Synthetic chemists lean on it to design, modify, and learn about chemical transformations. Its predictable performance lets teams run controlled experiments. During collaborative projects, I’ve watched postdocs evaluate how subtle changes in the amide structure can lead to different properties in test compounds. The reproducibility of N-Methylacetoacetamide helps researchers avoid chasing their tails due to inconsistent chemistry, which matters for advancing any field built on evidence.
Every industrial and academic lab faces tighter environmental standards. This chemical, while useful, prompts questions about waste and sustainability—challenges every chemist must grapple with. Solvent choices, storage, and byproduct handling all demand attention. I remember the careful tracking required during inventory audits and waste collection days. The push for greener synthesis has led some researchers to explore bio-based substitutes or recycle techniques. In some pharmaceutical companies, staff experiment with water-based reactions or employ catalysts that reduce hazardous output.
The story around N-Methylacetoacetamide isn’t only about what it does today, but where it can take industry and research tomorrow. Unlocking safer, more sustainable manufacturing will push everyone to rethink how these compounds fit into bigger frameworks. From my experience, hearing the voices of process engineers, junior chemists, and environmental officers during review meetings reveals a shared goal: advance what’s possible while keeping safety and responsibility front and center.