Few people outside the world of organic chemistry recognize N-Acetoacetyl-4-methylaniline, though its roots stretch through decades of innovation in both fine chemicals and dye production. Years ago, the push for new colorants and intermediates pushed research teams to engineer arylamine derivatives with functional groups tailored for specific reactivity. Early scientific reports detailed how introducing an acetoacetyl group onto a methyl-substituted aniline backbone helped bridge the worlds of aromatic chemistry and classic enolate-style reactions. The journey of this compound runs parallel to the movement toward more refined pigment synthesis throughout the twentieth century. Patents from the mid-1900s confirm how foundational these intermediates became as industrial applications multiplied, especially for pigments needed by textile and coating manufacturers. Knowing this history, I see N-Acetoacetyl-4-methylaniline as a prime example of how creative tweaks on a basic structure open new worlds for practical use.
Take the benzene ring—a workhorse in organic synthesis—add a methyl group at the 4-position and tack on an acetoacetyl group. That’s what you get here: a molecule with both aromatic stability and the reactive site from the acetoacetyl chain. In practice, it forms as a solid that behaves predictably under standard lab conditions, so storage and routine handling ask for few surprises. Its solubility and melting characteristics line up with many similar aromatic amines, making it manageable in both small-batch labs and larger processing environments. What matters most is the unique blend of properties: the sturdy ring, the electron-rich methyl push, and the keto-enol possibilities of the acetoacetyl tail. This combination shapes how it reacts and where it finds a home in industry.
Specifications for N-Acetoacetyl-4-methylaniline reflect its specialized role. Chemical purities run above 98% for research and industry, which protects downstream reactions from contamination headaches. Labels on commercial stocks spell out things like batch number and date, because traceability could become key if a pigment batch or drug precursor develops issues. Precise formulations matter most in R&D or regulatory submissions, where impurities or variant physical forms ripple down a chain and change an end result.
Preparation starts from p-toluidine, a simple and affordable aromatic amine. Chemists run acetoacetylation reactions—typically using diketene or acetoacetic ester—to install the acetoacetyl group on the nitrogen atom. The magic isn’t in fancy or esoteric reagents, but in steady conditions: controlled temperature, careful pH adjustment, and attentive workup to yield a clean product. This technique, honed over generations, avoids complicated purification steps. Researchers often leave open the option for small tweaks, swapping solvents or adding minor co-catalysts to raise yields or limit side products.
In old catalogs and new, N-Acetoacetyl-4-methylaniline appears under a range of names—sometimes as 4-Methylacetoacetanilide, or 4-Methylphenylacetoacetamide. Different fields and countries latch onto different synonyms, which can tangle up new students and old-timers alike. Mislabeling or mix-ups can slow research or prompt regulatory hiccups. That’s why chemists and industry pros push for IUPAC names or consistent labeling, helping keep projects running smoothly even in multinational collaborations.
Jobs involving aromatic amines or acetoacetyl derivatives call for respect and preparation. Those familiar with compounds like N-Acetoacetyl-4-methylaniline know that gloves, fume hoods, and splash protection aren’t for display—they shield the skin and lungs from unwanted exposure. Years of industry practice have cemented the routine: weigh powder with care, avoid open flames, and store away from moisture or excessive heat. Tight operational protocols echo recommendations from agencies like OSHA and REACH, bringing real-world consensus about what practices shield people and processes from harm.
The most famous uses remain in pigments, especially for producing vivid yellow colors and diazo-based paints or inks. Factories feed N-Acetoacetyl-4-methylaniline into complex dye formulations for textiles and plastics that need both brightness and stability. Lab scientists value its versatility as an intermediate for custom synthesis; its enolizable aceto group lines up for Michael additions, while the aniline nitrogen joins classic coupling reactions. In drug development, it functions as a building block, letting pharmaceutical chemists stitch together bigger, bioactive structures. Even outside these flagship sectors, new application experiments bubble up—for example, some teams test it in polymer modification or as a chelation agent in analytical chemistry.
Every time a compound like this enters a new application, fresh questions appear about its health or environmental impact. Toxicology studies chase evidence about metabolic byproducts, hoping to avoid the lessons learned from past aromatic amine mishaps. So far, reports suggest cautious optimism: available toxicity data for close relatives point toward manageable exposure risks when handled in professional settings, though some breakdown products have drawn scrutiny for possible mutagenic effects. Environmental concerns focus on proper treatment of industrial effluents—making sure that spent chemicals don’t slip into groundwater or rivers. As green chemistry gains ground, teams keep hunting for benign routes of synthesis and safer derivatives. I see promise in real-time monitoring and predictive models, which offer hope to both chemists and regulators for staying a step ahead.
Looking ahead, N-Acetoacetyl-4-methylaniline sits at the intersection of multiple research frontiers. Experts want new pigment molecules that resist fade and chemical attack, feeding interest in custom-tailored intermediates like this one. Synthetic chemists push the envelope by dreaming up new ligands, organocatalysts, and specialty polymers—often starting from time-tested molecules. Industry stakeholders know that tweaks to regulatory or supply chain rules could reshape practical use, so they follow updates in REACH or TSCA closely. As both green chemistry and circular economy models mature, we can expect sustainable production methods to rise in importance, coloring the future of this and similar specialty chemicals. The real value of N-Acetoacetyl-4-methylaniline doesn’t rest solely in its established pedigree, but in the creative potential it opens for tomorrows’ technological and environmental goals.
Long chemical names like N-Acetoacetyl-4-methylaniline might look intimidating, but chances are, products built on these compounds end up closer to home than most people expect. Many pigments and dyes that go into inks, textiles, and paints use substances like this as building blocks. My own kitchen drawer holds a rainbow of markers, and I’d bet a few rely on stable pigment cores derived from chemicals in this family. It’s easy to overlook the steps between a chemical plant and a dry-erase board, but that journey matters a lot.
N-Acetoacetyl-4-methylaniline fits into the machinery of industry by helping create pigments called azo dyes. These are colors you see in printed fabrics, colored plastics, and sometimes even food packaging. Azo dyes matter because they give products bright, lasting color, and their reliability turned them into a pillar of multiple industries. The process wouldn’t work as well without stable intermediates like this one. Years ago, I guided a factory tour where engineers explained the tight controls needed over every ingredient—nothing left to chance, since mistakes cost real money and sometimes risk safety.
As a writer with a long-standing curiosity about the nuts and bolts of manufacturing, I can’t help but appreciate how often innovation starts with someone tweaking a molecule to get a more vivid red or a fabric that doesn’t fade in the laundry. N-Acetoacetyl-4-methylaniline stands out for its stability and how it lets chemists create a huge palette of colors. This might not seem like a big deal except when you see the bottom line at a print shop or the frustration when a favorite shirt fades after a single wash. Every link in the supply chain counts.
Like many chemicals, concerns about human and environmental health ride in the background. Most azo dye intermediates raise questions after scientists flagged some related compounds for toxicity or persistence in wastewater. I’ve seen changes pushed by environmental advocates which led factories to invest in wastewater treatment or research safer replacements. While N-Acetoacetyl-4-methylaniline doesn’t carry the same baggage as all of its relatives, people working around these chemicals need to respect the risks. Companies have to invest in worker safety and responsible disposal. Europe and parts of Asia take a tougher stance on chemical safety, and trade partners watch standards closely.
One lesson I keep seeing repeated: safer production starts with better knowledge and real incentives for change—not empty regulations or paperwork. Research into greener dye synthesis and less hazardous intermediates ramps up every year. Some universities and companies explore plant-based alternatives, and the push for more regulated supply chains means companies track what’s in their products much more closely than before. Consumers asking for safer dyeing processes help drive that change, even if it’s just picking brands that talk about how their clothes are made.
In the end, specialty chemicals like N-Acetoacetyl-4-methylaniline play a quiet but important role in modern manufacturing. Their influence stretches from a chemist’s notebook to the aisles of a supermarket and on into the recycling stream. Every step toward safer, more transparent use of ingredients like these lifts the quality of goods and the health of communities around the globe.
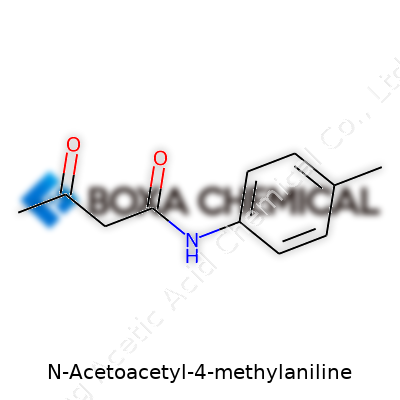
N-Acetoacetyl-4-methylaniline sounds technical, but its chemical structure holds the key to understanding why it matters. At its core, this molecule brings together a bit of aniline—known for its role in dyes and pharmaceuticals—with a twist: a methyl group on the fourth carbon of the benzene ring, and the addition of an acetoacetyl group on the nitrogen. That setup shapes both its properties and its significance in the world of dyes, pigments, and specialty chemicals.
Having studied chemistry and handled my share of aromatic compounds, I know how structure dictates behavior. Take the base: aniline, a benzene ring with an amino group. Add a methyl group on the “para” position, that’s 4-methylaniline (sometimes called p-toluidine in labs). Now, let’s acetoacetylate it—attach an acetoacetyl group (which looks like CH3COCH2CO–) to the nitrogen atom. This extra chunk changes everything: reactivity, solubility, and how the molecule interacts with others. The structure looks like this: the aromatic ring anchors the molecule, the methyl group gives it a unique electronic profile by making the ring more electron-rich, and the acetoacetyl group sets up sites for further reactions, especially in dye synthesis.
In manufacturing, minor tweaks to a molecule can mean a world of difference. N-Acetoacetyl-4-methylaniline plays its part in color chemistry. Its structure lets it serve as a coupling component for pigment and dye creation, especially for certain azo dyes. Azo dyes have colored textiles for decades, and a lot hinges on how the molecules react. By bringing acetoacetyl and methyl groups together on the ring, manufacturers set up the compound to combine with diazonium salts and create vivid, lasting colors. Factories rely on that structural predictability—anything that reduces batch variability keeps costs down and results up.
Handling and synthesizing aromatic amines, especially those modified like this, takes care. The acetoacetyl part can act as a weak acid and also take part in condensation reactions. Those features make it easy to react, but that can also raise safety flags—chemists know the risks of exposure. I’ve always worn gloves and made sure the fume hood worked, because some aniline derivatives can be absorbed through the skin or are volatile enough to risk inhalation. That’s not unique to this molecule, but it shapes every step between lab and large-scale production. Good ventilation, strict handling policies, and regular health monitoring cut risks down meaningfully.
The response to growing sustainability demands gives chemists a real challenge. The next steps lie in tweaking the synthesis process—less waste, safer solvents, better yields. Research in the last decade points toward greener acetoacetylation methods, often using catalytic processes or renewable starting materials. More widespread adoption of these approaches could shrink the environmental footprint of the pigment and dye industries without sacrificing quality or color strength. What we do in the lab ripples out to everything from medical diagnostics to the shade of a cotton t-shirt.
Looking at N-Acetoacetyl-4-methylaniline’s structure tells a lot about its potential and the responsibility involved with its use. Each tweak at the atomic level sends ripples through entire industries. Paying close attention to structure, safety, and sustainability ensures this molecule will hold its place in chemistry’s toolkit, both now and long into the future.
A lot of folks working with chemicals like N-Acetoacetyl-4-methylaniline eventually learn that trusting your instincts isn’t enough. Lab work isn’t forgiving when it comes to carelessness. I remember opening a bag of a new powder with no gloves—one whiff, one brush of dust, and I learned real quick why people repeat those safety tips. This compound doesn’t give second chances either. It finds its way in through your skin, your nose, or your lungs, and once exposure starts, you can’t undo it.
N-Acetoacetyl-4-methylaniline looks unassuming, like a lot of industrial chemicals, but exposure brings real risks. I always suit up: thick nitrile gloves, a proper lab coat that covers arms completely, and goggles that fit snug. Forgetting any one of those steps practically invites trouble. Even a small splash or a puff of dust can linger on your skin or irritate your eyes for hours. People sometimes think safety precautions are there to slow them down; I see them as a way to stay at work tomorrow and the day after, too.
Chemical fumes don’t just smell bad—they attack quietly. In stuffy corners of a lab, skipping the fume hood or ventilation system isn’t an option. I once underestimated a chemical until my throat burned for half a day. Now, I check the airflow and use hoods every time. N-Acetoacetyl-4-methylaniline needs respect for this reason: breathing it in can irritate airways, and long-term exposure can set up worse problems.
It pays to think ahead about spills. The compound stains and lingers on surfaces, so I use trays with raised edges and double-check for loose lids. Secure storage sounds simple, but one misplaced jar turns a shelf into a hazard zone. Keeping it in a cool, dry place, in shatter-proof containers, keeps things tidy and keeps accidents away. In my labs, spill kits and neutralizing agents aren’t buried on some back shelf; they’re close at hand where the action happens.
Hand washing isn’t just for surgeons. After handling chemicals, I scrub up every time—no shortcut. Any contact with N-Acetoacetyl-4-methylaniline means giving skin a real clean, not just a splash of water. I treat every towel and bit of used protective gear with the same caution. Thrown in the wrong bin, chemical waste causes headaches for waste handlers, so labeled hazardous waste bins are the only place it goes.
Learning these habits doesn’t happen on the first day. Proper training with a mentor made all the difference for me. That’s how I picked up tricks like recognizing skin sensitivity early and keeping the workspace organized. Peer-reviewed sources and safety data sheets stay open near my workstation, and I update myself whenever regulations or best practices change. Staying on top of protocols comes from community, not just rules on paper.
Accidents shrink when workplaces invest time in real training, visibility of hazard signs, and good communication across teams. I’d like to see more labs foster a culture where people look out for each other, point out missed steps, and keep safety gear in good shape. Chemical safety isn’t driven by fear. It’s built on experience, small reminders, and habits that protect lives and futures.
Chemistry sometimes looks like a world built on tiny numbers and complex formulas, but the sharp-eyed buyer, formulator, or researcher pays equal attention to the words tucked into a product’s fine print: purity and specification. Take N-Acetoacetyl-4-methylaniline as an example. This compound pops up in the dyes sector and also in custom synthesis work. People diving into its supply chain often ask, “How pure is this stuff, and what does its specification really say about how it will perform?” From my own days elbow-deep in the lab, reaching for chemicals with questionable paperwork lingers as a major source of headaches.
For N-Acetoacetyl-4-methylaniline, purity usually means talking about a percentage—often somewhere above 98%. But not all high numbers guarantee a worry-free experience. Purity says a lot about what’s missing rather than just what’s present. Extra bits—residual solvents, unreacted starting materials, or even moisture—can crash the whole operation, leading to unreliable results or bad performance in downstream applications. Small levels of impurities become magnified in sensitive dye chemistry. As someone who’s scrambled to troubleshoot off-color batches, I know these little mistakes slip past the eye easily unless you get firm documentation and honest reporting.
Manufacturers usually supply a specification sheet, and it’s tempting to skip straight to the assay line. That’s where purity by percentage gets listed. Digging deeper, smart teams also read about melting point, appearance, moisture content, and residual solvent limits. I’ve seen batches labeled as “white to pale yellow powder,” but subtle color shifts can hint at oxidative degradation. Moisture creeps into shipments thanks to poor packaging, and that small detail brings chaos in processes that need dryness. Trace metals or organic impurities, left from synthesis or poorly washed reaction vessels, also turn up if tests like HPLC, GC, or ICP get used with real effort. For N-Acetoacetyl-4-methylaniline, any reputable spec will include melting point—somewhere around 80-85°C—alongside that all-important purity.
Anyone serious about chemical manufacturing knows certificate of analysis (CoA) is worth its weight in gold only when it matches independent checks. I’ve seen shortcuts from intermediary suppliers who pass off incomplete data sheets or make up test results to keep prices low. Those bargains usually cost more after mistakes compound. There’s wisdom in sticking with sources who maintain transparent audit trails and welcome third-party verification. Quality outlasts rock-bottom pricing, especially when customers or regulators start asking questions.
Instead of treating spec sheets as a one-off procurement box to tick, it makes sense to form a relationship with suppliers who treat every order like a promise. Open requests for expanded impurity profiles or current good manufacturing practice (cGMP) alignment put the power in the buyer's hands. In my experience, those who develop these relationships find fewer ugly surprises.
Chemicals like N-Acetoacetyl-4-methylaniline don’t exist in a vacuum. Pressure to standardize reporting, use validated testing methods, and document everything clearly will only increase. I encourage labs and producers to demand transparency, not just accept a PDF full of numbers. Every user—from academic research to commercial scale—benefits when the purity and profile of a chemical isn’t a guessing game.
Not every chemical makes headlines. N-Acetoacetyl-4-methylaniline flies under the radar, but that doesn’t take away from its potential risks. I’ve spent years working with specialty chemicals, and I’ve seen what can unfold when the basics—like proper storage—get shrugged off. Once, a simple lapse in storing a chemical with similar properties led not just to a trashed sample, but to a ventilation system overhaul and a week of lost work for the lab. Small choices anchor big outcomes.
Aniline-based compounds share the same quirks: air, heat, and even a trace of dampness can cause changes. Water vapor sneaks through tiny cracks. I’ve learned that no storage space is ever totally perfect, but a cool, dry, and well-ventilated spot beats the alternatives hands down. Skip the windowsill and pass on shelving above heat ducts. Chemicals stored on high shelves near the ceiling end up hotter, sometimes by a good ten degrees compared to a lower spot—enough to speed up breakdown for some compounds.
It’s easy to think storage just needs a decent label on a shelf. But once, during a routine check, we found a bottle labeled perfectly but sitting right next to a strong oxidizer. That makes for a risky combination nobody wants. Storing incompatible chemicals side by side can trigger reactions even if containers look sealed. For N-Acetoacetyl-4-methylaniline, standard advice points toward keeping it away from bases and strong oxidants. Simple, sure, but shortcuts add up over time.
Most incidents I’ve seen start with the wrong kind of container. Glass with a solid screw cap works in many situations—unless someone loses the cap or forgets to tighten it after use. One summer, we found that even mild sunlight changed the color of a chemical sample, hinting at breakdown. All it took was an uncovered window and a week of afternoon light. Opaque or amber bottles help, especially when sunlight could be an issue, and using containers built to resist leaching keeps chemicals from picking up impurities.
Rules help, but good habits do the real heavy lifting. Locking up chemicals like N-Acetoacetyl-4-methylaniline in designated cabinets, keeping storage spaces free of clutter, and making regular checks part of a routine save trouble later. Posting clear reminder signs at storage spots sounds basic, but even seasoned chemists get distracted. Real safety culture grows from small actions repeated day in and day out—checking the seals, monitoring for spills, logging every use. Investing a few minutes during each shift could mean sparing hours of clean-up and investigation after an accident.
Plenty of regulations and safety data sheets spell out minimum requirements, but experience fills in the rest. The goal isn’t to lock away every substance, but to balance usefulness with risk. Handling N-Acetoacetyl-4-methylaniline responsibly keeps colleagues safe, labs running, and reputations intact. The time spent double-checking conditions or explaining storage to a new team member—those moments rarely make the news, but they build a safer, smarter working environment for everyone.