Anyone who’s spent enough hours in a lab with handfuls of reagent bottles might already recognize 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone, especially those who have followed the evolution of analytical chemistry for water, dyes, or even clinical biochemistry. This compound didn’t pop up out of nowhere. Its roots sink back almost a hundred years, growing from early work exploring pyrazolone derivatives for their strong reactivity and ability to create highly visible color changes. The drive for reliable colorimetric determinations set the stage; scientists at the dawn of the 20th century pushed for more precise ways to detect even trace amounts of substances, putting reagents like this in high demand. The chemical structure’s reliability made it a preferred choice for innovative reagent development in research hubs across Europe, and soon it became a regular item in the global laboratory toolkit. Through decades, as more advanced spectrophotometry and more standardized test methods emerged, so did modifications of this compound, which led to new applications far beyond its earliest uses.
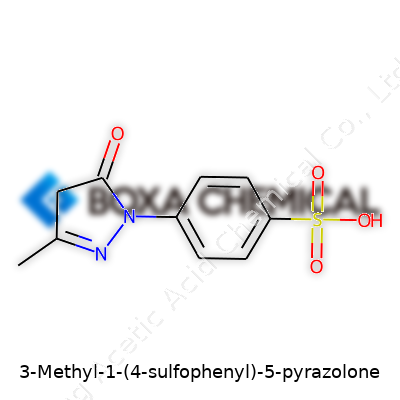
3-Methyl-1-(4-sulfophenyl)-5-pyrazolone stands out for its unique structure: a pyrazolone ring anchored by both a methyl and a sulfophenyl group. This touch of chemical engineering lends it properties that have found consistent use from small academic labs to the industrial giants. Many chemists keep a bottle on the shelf for pivotal color reactions—one of the classic tools for measuring phenols, in particular. Its high reactivity with certain ions and clear visual outputs make it accessible even for those who can’t invest in complex instrumentation. There’s a reason this compound appears in standard test protocols, and it’s mostly because it works—with a mix of reliability, accessibility, and a strong pedigree of scientific trust.
The powder form tends to appear as a pale yellow to orange solid, dissolving readily in water, thanks to the sulfonic acid functionality. That sulfonic group doesn’t just bolster solubility—it also helps minimize interference from other common contaminants, as I’ve seen time and again in side-by-side trials with less specialized reagents. In aqueous solution, you get a fine balance: stability over a wide pH range, and a reaction path that avoids annoying byproducts and messy precipitates. Chemically, the structure resists easy breakdown, meaning it sticks around long enough for extended analytical runs but doesn’t linger as an environmental pollutant in waste streams, at least not when handled with care.
Lab prep work pushes you to read labels with a sharp eye. Here, you see the systematic chemical name, alongside other identifiers, so confusion stays minimal. Purity levels can make or break an analysis; higher grades offer more consistency in titrations or colorimetric analysis. True, technical literature helps—manufacturers detail melting points, solubility in various solvents, and best handling temperatures. You don’t find confusing jargon on the label, just plain facts. This helps a technician cut through ambiguity, avoiding time lost to troubleshooting or detective work when a run doesn’t work as planned.
Old-school chemistry insists on careful synthesis and purification. The basic pathway to 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone involves the reaction of methylhydrazine with an appropriate sulfonated benzoyl compound, under mild conditions to avoid breaking the delicate ring structure. Isolation often uses crystallization or solvent-based separation to trim away impurities. This type of prep may look intimidating to the beginner, but it exemplifies classic organic synthesis: it’s all about balancing heat, pH, and careful pipetting. Oversights here ripple out—lower yields, contaminated batches, wasted resources—so the process rewards attention to detail.
In hands-on use, the most intriguing facet involves the way 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone interacts with phenolic compounds and metal ions. The resulting colored complexes stand out, delivering robust signals for detection—all without heavy instrumentation. In analytical circles, minor modifications of the structure unlock even greater sensitivity or specificity, and research papers regularly document tweaks for particular niche assays. Chemical versatility keeps it in rotation for custom synthesis projects, allowing research teams to chase down new targets by adjusting the substituents, or to tailor solubility for special solvents. The willingness of the compound to accommodate change cements its longevity and relevance.
Ask a dozen scientists about this compound, and you might hear several names. Long chemical handles like “4-Sulfo-phenyl-3-methyl-5-pyrazolone” or abbreviations such as MSPP come up, depending on the text or the lab. Documentation sometimes cycles through a stack of synonyms, so it matters to recognize these on supply lists or in archives. Recognizing these alternate names helps avoid confusion, especially when troubleshooting an assay based on articles written decades apart or halfway across the planet. Naming conventions might seem trivial, but a missed synonym in a research search costs hours or even days in lost results, making this an essential bit of practical lab wisdom.
Handling chemicals with unknown toxicity always deserves respect, and this pyrazolone derivative offers no exception. Actual hands-on work happens with gloves, eye protection, fume hood if needed—basic but non-negotiable. Storage conditions, spill response, and proper labeling rely on staff not cutting corners. I’ve seen what happens when attention lapses: subtle contamination, ruined baselines, sometimes even angry phone calls during audits. Material safety data sheets urge caution, particularly for worker exposure or environmental discharge. Keeping up with training, encouraging a safety-first mindset, and supporting whistleblowers for lax practices ensure trust in the lab, for staff and downstream users.
This compound pops up everywhere anyone analyzes phenolic wastewater, tests industrial process streams, or pushes for more accurate medical diagnostics. Whether someone’s monitoring dye industry discharges or running enzymatic assays for clinical markers, the reagent steps up with reliable color change and quantifiable outputs. Universities keep it close for teaching complexometric titration, letting the next generation see chemical theory in action. Environmental labs count on it, too, trying to juggle regulatory thresholds with sharp timeline pressures. Even as methods evolve, the core chemistry provides a safety net, ensuring accurate, repeatable measurements without demanding massive hardware budgets. That’s part of why demand hasn’t faded, despite talk of automation and AI-assisted diagnostics.
Advances in analytical chemistry fan the flames of curiosity, with research teams routinely testing modifications of 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone to sharpen accuracy, cut costs, and improve environmental compatibility. The old hands pass on the lessons: sometimes the best breakthroughs involve tweaking a known backbone or finding a more sustainable route to the same product. I’ve seen teams at both startups and academic centers experimenting with new derivatives to detect a broader range of substances—from heavy metals to drug metabolites in blood, where sensitivity and selectivity make the difference between success and failure. In the spirit of continuous refinement, the literature swells with improved protocols and stories of surprising results from modest chemical edits.
Decades of usage have sparked many toxicity studies, especially after deeper consciousness of occupational hazards gained steam in the late 20th century. While acute toxicity counts as low compared to many classic industrial chemicals, repeated exposure or careless disposal can provoke irritation, especially to skin or mucous membranes. Chronic exposure risks need more study, particularly since the breakdown products or combinations in complex matrices sometimes stray from well-understood metabolite pathways. That pushes for tighter oversight and more extensive data collection—don’t just rely on last year’s safety profile, stay vigilant and keep the conversation open with safety officers and environmental experts. Resilience in lab safety culture means acting on new evidence, not tradition.
Chemistry never stands still, though legacy reagents like 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone still manage to keep their seat at the table. New fields like green analytical chemistry ask serious questions about environmental persistence, solvent use, and potential for non-toxic breakdown. Eventually, researchers will push either for cleaner analogs, more robust lifecycle data, or hybrid systems mixing classic and emerging reagents. Continual tinkering—switching out functional groups, crafting more biodegradable derivatives, adopting closed-loop recycling for process chemicals—already shapes the next generation of standards. With pragmatic development, science keeps moving forward. Chemists build on what works, but push for better, safer, and more sustainable answers, not just for the sake of change but because they have seen firsthand the cost of stalling. This is how an established compound stays relevant—adaptation, scrutiny, and a culture that never stops asking how we can do the job better.
Ask someone on the street about 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone and you’ll probably get blank stares. The compound comes off a bit like a tongue-twister from a chemistry textbook. But for those working in labs or industries tied to colors and analysis, this unassuming molecule means business. I’ve spent time in a chemistry lab, and every so often, a tool stands out not because it makes headlines, but because it quietly gets the job done.
This pyrazolone derivative pops up most often as a dye intermediate or coupling component. In plain English, it helps create azo dyes—some of the most widely used colors for textiles, inks, and leather goods. Azo dyes made with these pyrazolones bring rich tones and lasting hues. There's a big demand for clothing and printing that keeps its color after countless washes, and research keeps pointing back to molecules like this one as the foundation. Based on published data, over half of dyes made for textiles around the globe belong to the azo family.
Manufacturers rely on these chemicals not just for their vivid color, but also for their chemical stability. If you’ve ever owned a t-shirt that faded quickly compared to one that stuck around for years, there’s a fair chance the better one leaned on more robust dye chemistry—likely involving intermediates like this.
There’s another life for 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone outside industrial vats: analytical chemistry. Lab workers often test for trace metals in water, medicine, or food supplies. Iron, in particular, has drawn a lot of research attention due to its nutritional importance and environmental impact. This pyrazolone compound teams up with iron ions to create a colored complex—one visible enough that even small amounts become easy to spot and measure. That kind of reliability means public health officials and water treatment specialists can act quickly if iron spikes to unsafe levels.
Telling from experience, clear and dependable chemical tests spare a lot of guesswork. Water analysis, where the margin for error feels razor-thin, gains a practical advantage from the straightforward detection that this compound offers.
No chemical comes without questions. The widespread use of dye intermediates raises concerns about what gets left behind during manufacturing or after disposal. Research into azo dye pollution has documented these substances showing up in rivers, affecting both wildlife and drinking water. Handling pyrazolone derivatives safely takes attention—not just from factories, but from watchdog organizations and regulatory bodies. The industry keeps searching for ways to lower the environmental cost, and shift toward greener alternatives where possible.
Drawing from scientific reports, efforts like improved wastewater treatment and stricter monitoring have helped, but communities living near industrial centers still report worries about exposure. I believe society benefits when researchers, regulators, and local residents sit at the same table to push innovation, share data, and stay vigilant about what flows into shared water sources.
3-Methyl-1-(4-sulfophenyl)-5-pyrazolone sits in the toolbox of countless chemists and manufacturers—powerful because it makes things happen, not because it’s famous. As textile and analytical labs keep evolving, the spotlight will keep shining on how to balance performance with responsibility. Investment into cleaner synthesis, combined with rising consumer awareness, can push dyes and reagents toward a future that keeps both color and conscience in check.
People who handle chemicals in labs or in industry usually want straightforward answers. No one likes surprises when it comes to safety. 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone, often discussed among analytical chemists and technicians because of its dye or reagent properties, draws some of these questions. Is it risky to handle this? What should you watch for in a practical sense?
Most research on this pyrazolone derivative focuses on its uses rather than its hazards. Yet if you scan through material safety data on similar sulfonated aromatic compounds, patterns emerge. Many of these substances can irritate skin and eyes. Some may create dust that’s unpleasant to breathe. Experience teaches that lab workers who skip the gloves or take shortcuts with goggles sometimes pay the price with skin rashes or watery eyes, which hurts productivity and can set back whole projects.
There’s another angle: pyrazolone derivatives have earned a bit of a reputation in toxicology. A number—including some related to pharmaceuticals—have caused trouble by prompting allergic responses in sensitive folks. So even without headline-making toxicity, basic mishandling can make for rough days.
Years of working with dyes and phenylpyrazolones have shown that people often underestimate low-level chemical exposure. Everyone remembers to watch out for acids and strong solvents. Less obvious compounds slip through because they seem less scary. This approach fails both individuals and workplaces because it ignores the slow, regular impact small exposures can have — chronic irritation, allergies, or worse.
Simple protective gear makes a difference here. Gloves rated for chemical splash, fitted goggles, lab coats, and well-ventilated benches usually prevent most incidents. Tidiness matters too. A little spilled powder or a fingerprint of colored dust might seem like a minor annoyance, but over time, such carelessness builds up risk. I’ve seen projects delayed not by dangerous reactions, but by staff needing weeks to recover from preventable skin irritation.
Products like 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone rarely trigger emergency alarms. No evidence ties such chemicals to long-term cancer risk at normal lab or industrial exposure. Agencies across Europe, the US, and Asia classify pyrazolone derivatives in ways that focus on basic hygiene. They require labeling for the possibility of irritation, not long-term systemic harm. This aligns with what most experienced chemists see on the job.
People sometimes hope regulations will outthink every risk, but rules only set a floor for safety. The standard practice of keeping chemicals in tightly closed containers, using fume extraction systems for powders, and cleaning up spills quickly does more for day-to-day protection than nearly any bureaucratic update.
In my view, the question “Is 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone safe to handle?” boils down to context and respect for the substance. With gloves, goggles, and a tidy workspace, the answer is as good as it gets for common chemical work. Lax habits and ignorance invite problems. Training helps, but so does humility — assuming a compound deserves care, even if it doesn’t seem deadly. That’s what really defines safe handling.
Every time I step into a chemical storeroom, the quiet hum of refrigerators and warning signs on cabinets remind me how much thought goes into protecting both people and the substances on those shelves. 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone, a name that doesn’t trip off the tongue, sits among those compounds with special needs. Researchers and lab technicians everywhere know it only takes one slip in storage standards to compromise safety or quality.
Like many substances used in research and industry, this chemical doesn’t take kindly to heat or moisture. Heat speeds up reactions in ways that aren’t always predictable, and water can cause clumping or even chemical breakdown. I remember a graduate class where a jar, just carelessly capped, ruined an entire batch because humidity seeped in overnight. We lost not just the compound but a week’s worth of work. Room temperature in a climate-controlled setting, dry air, and sealed containers become non-negotiable. These aren’t quirks, but lessons that come from experience and mishap.
Direct sunlight makes things worse. UV light from the sun can alter the chemical structure of organic molecules surreptitiously. If you’ve ever seen a faded label or warped plastic by a window, you’ve already seen what light can do. Compounds like this one need opaque bottles, tucked away in cabinets away from windows. A simple cardboard box on a shelf won’t cut it; laboratory safety guidelines back this up with decades of real-world evidence.
Contamination poses another risk. 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone interacts unpredictably with everyday lab dust, acids, or even the wrong cleaning agent left behind from a previous spill. I once watched a student handle a powder near an open bottle of bleach. Instantly, lab supervisors stepped in to point out the dangers of airborne cross-contamination. Keeping chemicals in separate, clearly labeled spaces may sound obvious, but it saves money and ensures experiments aren’t botched by unseen reactions.
Accidental spills or improper storage practices often carry bigger consequences than ruined research. Inhaling organic powders or sulfophenyl derivatives is a genuine health hazard. That’s not alarmist talk—lung damage, allergic reactions, or worse, all stem from simple storage mishaps. Anyone who’s ever had a coughing fit after a minor spill knows the value of keeping gloves and masks easily accessible, and of monitoring containers for cracks or worn seals. Regular audits and signage reinforce a sense of shared responsibility. It’s the little things—fresh desiccants in jar lids, double-bagging powders for transport, working in fume hoods—that keep a lab running safely.
Regulators like OSHA and scientific societies don’t just love paperwork; their guidelines are built on incident reports and best practices. Reading through safety data sheets, you see patterns emerge: store in a cool, dry place, away from acids or oxidizing agents, inside tightly closed containers. I’ve always found the most successful labs rely on a culture of vigilance and open communication. If someone isn’t sure how a chemical should be stored, people speak up instead of covering it up. Every seasoned chemist has a story about one “close call” that could have been avoided with just a little more attention to storage details.
The world’s labs and industries depend on small actions taken every day. That humility and care for proper storage, more than any innovation, keeps workers safe, reputations intact, and discoveries repeatable. 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone serves as just one example of why it pays to never treat chemical storage as an afterthought.
People outside chemistry see names like 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone and think, “More alphabet soup for lab folks.” For chemists and anyone dipping their toes into pharmaceutical or dye research, understanding the structure behind this name makes a real difference. This compound doesn’t just show up in obscure textbooks; it’s carved its mark in the history of chemical dyes and research chemicals. I still remember the first time my university lab mapped out its structure on the whiteboard, marker ink squeaking as we pieced together each part. That moment made scientific names real and useful.
Each chunk of 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone points to a corner of the molecule. “Pyrazolone” signals a five-membered ring with two adjacent nitrogen atoms and a ketone group, a core found in many pharmaceuticals and colorants. That distinctive “3-Methyl” means a methyl group—just a simple carbon group—hangs off the third carbon. The “1-(4-sulfophenyl)” adds a benzene ring, hanging from the first position, its own fourth spot sporting a sulfo group, which brings water solubility and reactivity.
Draw up the structure on paper and the puzzle comes alive: a pyrazolone ring with two nitrogens side by side, methyl off the third carbon, a phenyl group off the first nitrogen, sulfonated to let the compound dissolve easily in water. This layout gives the molecule some of its best known properties—reactivity, water compatibility, and the ability to form bright, stable color complexes with metals like iron or copper.
Understanding molecular structure isn’t about memorizing squiggles—it’s the gateway to new discoveries and real-world applications. Take medicine: small tweaks to this backbone have produced anti-inflammatory drugs and fever reducers. Chemists still reach for the basic 5-pyrazolone shape in the hunt for new therapies. In dye stuff, its vivid, metal-binding ability transformed the textile and paper industries, leading to lasting, bright colors that don’t wash out.
This compound even played a part in how analysts detect trace metals. Think of wastewater or food safety testing—labs use 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone-based reagents to pick up even faint amounts of heavy metals. The ring structure, armed with its sulfo group, clings to metal ions and produces a colored compound that shows exactly what’s in the beaker. These color changes build trust that a factory's effluent isn’t poisoning the river.
Chemical structures like this spark questions about safety and environmental impact. The sulfonated benzene ring delivers good water solubility, but industrial-scale synthesis can spill pollutants if manufacturing isn’t properly controlled. That gives chemists and regulators something concrete to improve. Green chemistry looks to make these processes cleaner, focusing on ways to limit harmful byproducts and nudge companies into using more sustainable solvents.
Better understanding, cleaner methods, tighter safety checks—these start with respect for complex structures. The more closely scientists and engineers look at each part of molecules like 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone, the more responsibly the world can use them in technology, testing, and beyond.
Every so often, a name like 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone pops up, and it sounds like something straight from a chemistry textbook. Anyone who’s worked in labs or industrial settings might know this compound shows up in manufacturing and scientific research. Tossing out unwanted chemicals, though, isn’t just a “put-it-in-the-trash” task—it’s a point where responsibility meets reality. After all, improper disposal often spells trouble for people and the planet down the line.
Every year, industrial waste sneaks its way into news cycles linked to polluted rivers, failing ecosystems, or health scares. Chemicals like 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone may not live in the headlines, but their story matters behind the scenes. They don’t just vanish; those molecules persist. Some break down slowly, others resist environmental forces for years. Pouring compounds like this down a drain or tossing them in the regular trash could send them into water systems or landfills, where they might disrupt local biology or push past filtration methods at a water treatment plant. These disruptions can hurt fish, unsettle the food chain, or, in unforgiving cases, find their way back to the dinner table.
Back in college, our chemistry team had a supervisor who drilled home that treating lab waste like kitchen garbage just puts more headaches on someone else. Leftover reagents from experiments got boxed, labeled, and sent to the hazardous materials crew—no ifs, ands, or shortcuts. The mantra: “If you know what’s inside, treat it like you care.” Habits from the lab translate into industry, with one big difference: scale. One beaker doesn’t do much, but barrels of waste add up quickly, and the ripple effect grows.
Safe disposal starts by keeping the stuff separate. Dumping everything together only makes sorting and treatment harder. Good record-keeping means knowing exactly what’s on hand, so there’s no mixing up chemical types. Labs and factories often turn to certified hazardous waste specialists for a reason: these pros don’t guess or move things out of sight—they follow serious rules set by environmental agencies.
Incineration sometimes gets a bad rap, but for organic compounds that refuse to break down, high-temp incinerators designed for hazardous waste can destroy dangerous molecules. It takes specialized equipment to manage the risks of toxic gas release, but with oversight, it works far better than letting the material seep away in the wild. Water treatment plants weren’t built with stubborn lab chemicals in mind. Regulations don’t come from nowhere—they’re a hard-learned lesson after cases of groundwater contamination or sickened fish.
Most organizations, from teaching labs to chemical factories, can partner up with local hazardous waste programs. The fees pay for safety, but the payoff means a lower risk of fines, cleaner water, and a real shot at limiting accidents. Even in smaller labs, simply looking up local waste collection events, or working with a university’s environmental health officers, can make a world of difference.
For anyone dealing with leftover 3-Methyl-1-(4-sulfophenyl)-5-pyrazolone, giving disposal the respect it deserves isn't about pleasing some faceless agency. It’s about protecting people before problems grow too big to fix. Simple steps—keep it separate, label it right, and hand it to professionals—show the world that handling our mess the right way still matters.