Curious minds in early organic chemistry labs started exploring derivatives of acetoacetanilide long before the pharmaceutical boom. 2-Methylacetoacetanilide surfaced on the scene when altering substitution patterns on benzene became possible with better catalysts and purification tools. Decades ago, industry shifted from simple benzenes to more complex functionalities, making this compound’s methyl group a valuable tweak for chemical behavior. I remember sifting through journals that highlighted how small structural tweaks created new possibilities in dyes, pigments, and drug intermediates. This compound, often noted for its subtle but crucial differences, marked a steady evolution in organic synthesis, reflecting how chemists always look for tiny improvements that yield big results.
For many, 2-Methylacetoacetanilide sounds like another long-winded lab chemical. Strip away the jargon, and this molecule plays a flexible role. Not only does it act as a chemical building block, but its core structure helps tailor molecules for specific end uses. It consists of a benzene ring bearing a methyl group and an acetoacetyl group linked with an aniline backbone. Scientists often seek these subtle modifications to create dyes for textiles, inks, and even certain pharmaceuticals. Factories that lean on organic pigments recognize how a single methyl group can shift properties just enough to offer richer colors or improved performance—even before talking about downstream chemistry.
Holding a sample of 2-Methylacetoacetanilide, you find a pale solid that melts easily in a warm beaker. The methyl group nudges its melting point lower than unsubstituted versions. Its solubility profile follows expectations: solvents like ethanol and acetone dissolve it, bringing flexibility to synthesis and formulation work. Where I’ve seen it most, this compound handles mild pH ranges well. It resists rapid breakdown under ordinary lab conditions, which helps when long reaction monitoring is needed. In terms of stability, the methyl group guards certain reaction sites, nudging synthetic chemists toward more selective modifications without extra defensive steps.
Most labels on bottles of 2-Methylacetoacetanilide run basic: chemical name, batch identification, and purity, usually topping 98%. Every time I’ve visited a production site, quality teams check moisture content, particle size, and absence of related impurities—a critical step before anyone trusts the material for downstream catalysts or pharmaceuticals. Documentation details handling precautions, ensuring the user maintains dry, cool storage. Many labs store it away from oxidizers and acids, and every label reminds the handler of these needs. These steps seem routine, but such habits cut down on waste, failed batches, or accidental degradation.
Preparing 2-Methylacetoacetanilide relies on the tried-and-true approach: acetoacetylation of 2-methylaniline. Mixing 2-methylaniline with diketene or an equivalent acetoacetylating agent under controlled temperature leads to the target compound. Often, I’ve seen labs use solvents like toluene or acetic acid to guide the reaction and encourage crystallization. Temperature, pH, and post-reaction purification define the final product’s quality. In scaling up, continuous flow setups bring better control, more consistent batches, and fewer side-products. These advances save resources, reduce emissions, and cut down manual labor, which seems small but makes a world of difference for both workers and the environment.
This compound stands out for its active methylene group. Chemists take advantage of this spot for condensations—transforming it into pigments or other functionalized molecules. The methyl group at the ortho position influences reactivity by providing some steric hindrance and modifying electron density, leading to more selective couplings or cyclizations. Over many syntheses at the bench, I’ve found that its selectivity often means cleaner products in fewer steps. The ability to tweak the acetoacetyl chain or substitute further on the aromatic ring unlocks new colorants or intermediates for pharmaceutical ingredients. Innovations using mild catalysts like palladium or green solvents keep improving how efficiently it transforms into more complex molecules—a trend that mirrors my own focus on making chemistry both cleaner and more cost-effective.
People in chemical circles call 2-Methylacetoacetanilide by its mouthful of synonyms. You’ll see names like o-Tolylacetoacetanilide, 2-Methylacetoacetanilide, and even 2-‘Methylphenyl’ derivatives scattered across older literature. Trade names appear in industrial catalogues and paint ingredient lists, sometimes disguised under codes. Translating between these takes practice—I’ve learned over time to always check the IUPAC structure instead of relying on trade or common names, since mistakes can trigger wasted time or regulatory headaches. Clarity in names ensures the right product lands in the right research, formulation, or manufacturing stream.
Working with acetoacetanilide derivatives brings standard laboratory safety responsibilities. 2-Methylacetoacetanilide, not classified as acutely toxic, still calls for gloves, eye protection, and proper ventilation during handling. Fine powders kick up dust, so mask use and localized ventilation systems earn their place in every workflow. Over years in shared lab spaces, I‘ve seen even straightforward chemicals cause minor trouble when folks relax about storage and disposal. Following national and regional chemical regulations, including tracking for hazardous waste streams, reduces risk both in the workspace and for the environment downstream. Posting clear safety sheets and hands-on training bridge knowledge gaps, which keeps teams and cleanup crews safer in the long run.
The main scene for this compound lies in the world of dyes and pigments, especially for textiles, leather, and industrial coatings. That methyl group, subtle though it seems, enables formation of brilliant yellow pigments widely used in fabric printing and inks. Formulators value the resulting shade stability and brightness. Beyond colors, the pharmaceuticals industry turns to this building block in drug discovery, leveraging its reactivity to produce intermediates for more complicated molecules. Restrictions on hazardous colorants pushed many manufacturers toward safer alternatives, making derivatives of acetoacetanilide like this one more important in regulatory-compliant products. Some research even circles back to biological sensors and specialty polymers, as the need for tailored materials keeps growing. Every time a new application appears—whether in an eco-friendly coating or a less toxic intermediate—it’s clear that small molecules still matter in shaping big trends.
Labs and industry alike keep tinkering with acetoacetanilide chemistry. Researchers keep optimizing the synthesis to lower waste, shorten reaction steps, and increase atom economy. Looking at modern literature, there’s a real push on green chemistry—using bioderived solvents or leveraging less toxic reagents in the acetoacetylation. I’ve spent months troubleshooting reactions, realizing that minor tweaks in temperature or reagent purity could shave hours or produce more crystalline yields. New analytical tools, such as advanced NMR and mass spectrometry, make it easier to spot and correct impurities early. Current projects dig deeper into expanding the range of final products—whether that’s a longer-lasting dye or a drug intermediate with better pharmacokinetics. These efforts show that even established chemistry keeps evolving as markets demand both safer and more sustainable solutions.
Toxicology studies for 2-Methylacetoacetanilide show a relatively low hazard profile in common laboratory settings. Acute toxicity remains low in rodent studies, but like similar anilide derivatives, there’s some caution around chronic exposure and breakdown products. Scientists have tested the compound for skin and ocular irritation, with results generally supporting safe handling with standard protective gear. I’ve found that most of the real risk comes from improper disposal or long-term environmental buildup, especially since aromatic compounds can persist in soil and waterways. Environmental strategies stress containment, treatment, and minimization in manufacturing waste. More data is always better—so ongoing research aims to clarify long-term ecological and occupational risks, especially as applications and production volumes climb.
Interest in 2-Methylacetoacetanilide remains solid as sustainable manufacturing and safer chemical building blocks take center stage. Advances in green chemistry promise cleaner, more energy-efficient production routes that reduce emissions and waste. Digitalization in manufacturing helps monitor purity, increase throughput, and cut down on resource use. As demand grows for brighter and more stable pigments in textiles, inks, and specialty applications, the need for specialty derivatives like this will not go away. Researchers are also pushing boundaries, using machine learning to predict new reactions and optimize existing syntheses, revealing opportunities to expand application spaces or uncover new, safer uses. This chemical, rooted in old-school organic labs, keeps finding itself renewed by new tools, new rules, and new markets—a testament to chemistry’s enduring drive for progress.
2-Methylacetoacetanilide rarely gets the spotlight outside of chemistry circles, but its impact shows up in places people might not expect. Someone sees the vibrant yellow on a street sign or enjoys a sunny shade on fabrics or paints — in many cases, compounds like 2-Methylacetoacetanilide played a role. I first heard about this chemical during an old conversation with a friend who dyes textiles, and the way she described parts of the dye process stuck with me. She said 2-Methylacetoacetanilide isn’t directly the final dye, but acts as a stepping stone to something vivid and useful.
This compound helps create azo dyes, a massive group of synthetic colors. By itself, 2-Methylacetoacetanilide sits on a shelf looking almost unremarkable — a white or pale solid, easy to overlook. During manufacturing, though, it takes part in chemical reactions that transform it into striking pigments. People probably appreciate the result every day, without knowing what went into it.
For most of the colors that industries rely on, durability matters. People notice when a shirt loses its brightness after a wash or a sign fades in sunlight. Compounds built from 2-Methylacetoacetanilide help create dyes that hold up under stress. Companies often choose these derivatives because they hold onto their boldness, and they work under a range of conditions — in hot textile vats, on coated metal, or in plastic goods.
Global statistics show that the use of synthetic dyes grew over the last few decades. Azo dyes make up more than half of modern dyes, with roots often tracing back to chemicals like 2-Methylacetoacetanilide. The market for organic pigments keeps climbing, reflecting demand from industries that care about appearance and performance both.
I’ve read stories from environmental scientists who track the fate of chemical dyes in rivers and streams near industrial zones. Pigments made from 2-Methylacetoacetanilide don’t pose much risk in finished goods, but things get complicated during manufacturing or disposal. Regulatory agencies stepped in several times to check whether specific chemicals in this group break down into anything dangerous, or stick around too long in the environment. Some azo dyes drew serious concern for their ability to turn into aromatic amines, which can cause health problems.
It makes sense that industries need to pay close attention to how these compounds behave before, during, and after use. Routine testing of water near dye factories turned up real problems in some areas, which forced companies to improve waste management and introduce safer processes. That effort continues today, as researchers hunt for alternative chemicals with a smaller footprint, or improve recovery systems so less dye escapes into ecosystems.
2-Methylacetoacetanilide isn’t leaving the scene any time soon. As demand for bold colors holds strong, people keep refining how these chemicals show up in the supply chain. Some newer factories already rely on closed-loop water systems. Others experiment with biodegradable dyes made from different building blocks, often inspired by natural pigments. Progress feels real but slow, and part of that challenge comes from ongoing demand for rich, consistent color across so many fields.
As the discussion grows louder around sustainable manufacturing, chemicals like 2-Methylacetoacetanilide deserve a place in the conversation. That means weighing the need for performance with care for public health and the environment, and making room for innovation that respects both.
I’ve spent plenty of time flipping through chemistry textbooks, hunting for answers to questions like “What is the chemical structure of 2-Methylacetoacetanilide?” On the surface, it sounds technical—but scratch that surface and you’ll notice how the structure tells a bigger story about function, safety, and history in both labs and manufacturing settings.
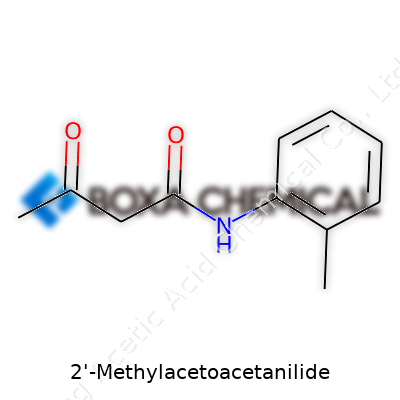
Break the name down, and the pieces make a pattern familiar to anyone who’s picked up organic chemistry. There’s a benzene ring—a foundational block in hundreds of everyday compounds. Out of that ring grows an amide branch, built from an aniline group. Tagging along is a methyl group at the number 2 spot (which just means it’s attached to the adjacent carbon on the benzene). The acetoacetyl bit adds another carbon chain with a double-bonded oxygen, so everything balances out with a chemical formula of C11H13NO2. Draw it out, and the shape starts with the benzene ring, linking through a nitrogen bridge to an acetoacetyl tail and sporting a methyl sprout. It’s less like assembling furniture and more like arranging Lego bricks to build something both targeted and sturdy.
Swapping in a methyl group does more than just add heft to the molecule. Methyl, small as it is, can tilt the scales on how the molecule behaves in various chemical reactions. In pharmaceuticals, dyes, and even some agrochemicals, minute tweaks like this change how things dissolve, interact, or resist breaking down. That methyl group, sitting at its spot, can make the difference between something benign and something with a touch of toxicity—or boost how a colorant bonds to fabric versus rinsing away in the wash.
Stepping outside the neat diagrams, I like to connect these facts with real-world results. Take quality control in manufacturing. The specific structure of 2-Methylacetoacetanilide means it might turn up in the quality assurance of dyes or drugs, because slight changes in structure could affect safety or effectiveness. It all comes down to knowing exactly what molecule you have on your hands, especially in regulated industries.
On the safety side, that extra methyl group can sometimes make a substance less stable or nudge it into a different hazard classification. Knowing the full shape of a molecule lets risk assessors predict how it will behave in the environment—will it degrade easily, or stick around?—and how it might affect human or animal health. Research shows even small structural changes can alter toxicity profiles, so details matter.
Managing substances like 2-Methylacetoacetanilide responsibly requires sharp attention to molecular minutiae. Labs use spectroscopy (NMR, IR) and chromatography to double-check that they’re dealing with the right form, not some impurity or cousin compound. Factories invest in monitoring every batch. Regulators often update guidelines based on new research into how structural changes impact safety.
Challenges remain, especially with keeping analysis thorough enough to avoid contaminants without driving up costs. One step forward could be wider use of rapid testing technologies, which give a real-time look at structure and purity before materials ever leave the plant. That puts the science into practice where it makes the biggest difference: in the hands of those who shape, use, and monitor chemicals every day.
2-Methylacetoacetanilide doesn’t turn up at dinner parties. It has a much humbler place, usually locked away in a research lab or production site. Those handling this chemical carry an unwritten contract: protect yourself, protect your coworkers, and protect your environment. Gloves and goggles aren’t just a dress code; they’re the real line of defense. I remember my first experience with chemicals of this class as a young assistant in a university lab—one poorly rinsed glass beaker nearly resulted in a ruined day and a chemical burn. These incidents leave an impression, teaching that familiarity breeds more risk than inexperience.
It doesn’t take a spilled beaker to realize that breathing fine particles or vapors could invite health problems. With compounds like this, exposure through skin or inhalation can be the start of a long story nobody wants to tell. I’ve worked alongside lab techs who would shake off safety reminders—until someone ended up with a nasty rash. The facts speak louder than any memo: chemical irritants don’t negotiate, and allergic responses don’t respect experience.
Industry standards always lean on chemical-resistant gloves, eye protection, and fume hoods. But practical knowledge suggests going beyond that checklist. For instance, I avoid open-toed shoes and stick with a sturdy lab coat that covers the wrists. All the safety gear in the world won’t help if you let clutter crowd the workspace or ignore a faint chemical smell. Airborne exposure can creep up quietly—so a well-maintained ventilation system deserves just as much respect as any lab instrument. If a spill occurs, it pays to know exactly where the nearest absorbent pads and wash stations are located, not just on a map but by heart.
Supervisors must guarantee everyone knows the drill before the first bottle opens—reading a label isn’t enough. I’ve seen robust safety cultures where teams rehearse spill scenarios and quiz each other on material hazards. This offers much more than formal training; it sets up a sense of community responsibility. If I spot a missing label or damaged container, I call it out. It’s far better to hold up production for a minute than nurse regrets later.
Disposal raises a different set of issues. My experience in labs has taught me never to pour leftover chemicals down the sink—a lesson often passed down from the most seasoned crew members. Local environmental rules exist for a reason. Flushing hazardous waste endangers water sources and pulls public health into the mix. Designated chemical waste bins, logged and kept secure, are the only option I trust.
If there’s anything decades in research environments have shown me, it’s that risk comes from taking shortcuts. Good ventilation, personal protective equipment, and regular training form the basics. Open discussion between colleagues keeps small lapses from turning into big problems. Handling chemicals like 2-Methylacetoacetanilide means accepting this responsibility every single day.
Understanding the molecular weight of a compound like 2-Methylacetoacetanilide isn't just an academic exercise—it plays a direct role in daily work in laboratories and manufacturing sites. The molecular weight, calculated as 191.23 grams per mole for this compound, comes from the precise arrangement of carbon, hydrogen, nitrogen, and oxygen atoms. Accurate knowledge of this value forms the foundation for preparing reagents, scaling up production, or simply conducting safe and efficient experiments.
I still remember my early days in a research lab, carefully weighing chemicals for organic synthesis. More than once, a single mistake in calculating molar masses set me back hours or ruined whole batches. Reproducible results depended on getting these numbers right from the start. No researcher wants to run a reaction with too much or too little of a reactant because the math led them astray.
Using the wrong molecular weight during chemical preparation means wasted time, wasted money, and greater risk of dangerous incidents. In industrial settings, the stakes climb higher. An incorrect batch can put a dent in a company's bottom line and erode trust with clients. It also impacts environmental safety. Over-ordering or under-utilizing chemicals ramps up waste and complicates disposal. The risks grow when regulations demand strict accountability for what is being used and discharged.
Since 2-Methylacetoacetanilide appears in pharmaceutical and dye chemistry, a miscalculation in its molecular weight—191.23 g/mol—could throw off entire syntheses. Pharmaceutical standards are famously unforgiving about dosing errors. Even a small misstep might affect patient safety or violate compliance standards.
Trustworthy results require double-checking every calculation. This means pulling out reference books or verifying with reputable online databases. It always pays to question a single source and seek peer-reviewed data. Chemical suppliers occasionally post varying values if a compound’s formula isn’t clear, so clarifying the molecular structure first removes confusion. For 2-Methylacetoacetanilide, looking up its formula—C11H13NO2—and breaking down the atomic weights explains the math: carbon (12.01) × 11, hydrogen (1.01) × 13, nitrogen (14.01), oxygen (16.00) × 2.
Access to properly calibrated balances in the lab helps but must be matched by the right values on paper. Computerized laboratory information systems help catch mistakes, though manual calculators or spreadsheets remain common backup tools. Documenting every calculation step prevents assumptions and helps others check your work if questions arise.
Sharing experience with new colleagues goes a long way. I’ve seen frustration on the faces of young chemists who thought mathematics was behind them once they walked into the lab. Truth is, accuracy in arithmetic keeps the whole operation moving forward. Showing others how to spot-check a calculation using different sources makes for a safer, more productive team environment.
Chemicals like 2-Methylacetoacetanilide don’t offer any room for guesswork. Adopting thorough habits grounded in science keeps projects on track and people out of harm’s way. Every number deserves respect, as lives and livelihoods depend on it.
Anyone who’s spent time in a lab knows sloppy storage can destroy more than a budget—it puts health and research at risk. 2-Methylacetoacetanilide doesn’t grab headlines, but the way it’s kept behind closed doors says a lot about a lab’s culture. This chemical pops up in everything from pigment and pharmaceutical research to some more specialized synthesis work. If you overlook storage basics, you invite spoiled samples, unpredictability, or even unwanted safety incidents.
There’s a story I always share with new assistants. A jar of a similar compound, left near a window because someone figured sunlight didn’t matter, changed color in a week. The analysis ran off track. All that time, effort, and cash—gone, just from lazy storage. 2-Methylacetoacetanilide is sensitive enough that moisture or too much heat cause changes researchers might not catch until it’s too late. Storing powders and crystals in closed containers, away from sources of excessive heat or moisture, isn’t overkill. It’s basic respect for your own work.
Consistent temperature matters most. Once, after storing similar organic compounds at room temperature, I saw degradation within months, even if at first nothing obvious appeared off. Chemical databases and reference guides generally suggest keeping 2-Methylacetoacetanilide at room temperature, well-sealed, and dry. Many labs use desiccators or those handy silica gel packs for an extra layer of confidence. Light exposure rarely helps. Too much sunlight definitely speeds unwanted breakdown. A cabinet or dedicated storage box with opaque sides makes a difference. These steps don’t just protect the original material—they protect anyone who uses it later.
Improper storage goes beyond one-off losses. Old, degraded samples introduce errors that nobody traces back to the root cause. Accidental inhalation and skin contact become more likely if containers sit open or humidity pulls particles into the room. Raising awareness and marking dates on containers makes sense, especially in busy spaces where turnover is high. It’s easy to forget what’s been sitting too long unless someone makes a habit of checking.
Research budgets shrink all the time, so practical methods matter. Locking hazardous chemicals in metal cabinets with fans or simple desiccant tubs prevents most disasters. Labeling containers clearly avoids guessing games about freshness. For smaller stocks, keeping records of batch numbers and dates helps track stability without fancy tech. Some teams add a layer of accountability by assigning routine checks, much like a grocery store rotates milk. Nothing fancy—just solid habits.
Some folks say storage is mundane. They’re wrong. The quality of a chemical can pivot on days of bad storage, especially for anything bound for synthesis or analysis. Faulty handling isn’t just a paperwork headache; it can trigger health risks for team members. The labs that take storage seriously usually see fewer surprises—good for people, pocketbooks, and actual progress. It’s the small stuff, like the way you store a bottle in June, that shapes next year’s breakthroughs.