People have been tinkering with aniline derivatives for over a century, chasing new colors, crop protections, and pharmaceuticals. 2-Chloroacetoacetanilide is one of those quiet, workhorse compounds born out of that era when chemists hunted for simple but powerful building blocks. There’s no single eureka moment behind its discovery; it slid naturally from earlier work on acetoacetanilide derivatives, fueled by both curiosity and the demands of the dye industry. Chlorine’s addition to the acetoacetanilide backbone opened doors that plain acetoacetanilide just couldn’t unlock. The early to mid 20th century saw an uptick in patents and technical papers as industries realized this compound could handle tasks ranging from pigments to pharmaceuticals. My respect for those early tinkerers only grows with every new application that emerges.
What catches the eye about 2-Chloroacetoacetanilide is not its glamour but its utility. This white or off-white crystalline powder rarely gets public spotlight. Yet, behind the scenes, it serves as a key intermediate in manufacturing brighter colors and better agrochemicals. Its structure — a chloro-functionalized acetoacetanilide — sets it up for a lot of further chemistry, making it a favorite among technical teams looking to make more complex molecules. Firms specializing in pigment or pesticide intermediates keep it on their shelves, trusting in its reliability for batch-to-batch performance. Its name might get lost in the blur of industrial catalogues, but without it, many fields would have to scramble for alternatives.
Chemists respect 2-Chloroacetoacetanilide for its distinct melting point, reasonable stability, and moderate reactivity. It doesn’t have the volatility of a troublemaker, letting chemists concentrate on reaction outcomes rather than worrying about runaway processes. Its solubility and crystalline nature help with both storage and further processing — fewer surprises in the drum mean fewer headaches in production planning. The compound boasts a unique ability to participate in carbon-carbon and carbon-nitrogen bond formations. Anyone who has spent time fighting solubility or stability gremlins in a plant will appreciate this material's relatively straightforward handling profile.
Quality-minded producers focus on the purity and moisture content most of all. Slight impurity spikes can foul up downstream reactions, so a reliable product needs clear specs for minimum assay and limits for related substances. Labels must communicate hazard statements boldly, with the usual attention to handling chloro-organic compounds. I’ve seen too many production line slowdowns caused by unclear packaging or missing batch numbers, so good labeling here isn’t about compliance — it’s about keeping things running and workers safe.
Practically, the main route involves acetoacetanilide reacting with an appropriate chlorinating agent, often under controlled temperatures and anhydrous conditions to keep by-products in check. A well-designed synthesis needs careful balance between reactivity and selectivity. Even a moment’s distraction, especially in scaling from lab flask to industrial reactor, can throw off the yields or saddle a batch with side products. Operators value replicable methods that use accessible reagents and don’t saddle plant managers with waste disposal nightmares. One thing I’ve learned: the real challenge is translating a promising lab recipe into a vessel-friendly process that meets industrial demand without blowing up the waste budget.
The chemical backbone of 2-Chloroacetoacetanilide offers an inviting site for nucleophilic substitutions and condensations. It functions as a stepping stone in synthesizing a web of other products. Chemists take special care when tweaking the chlorine group since it changes the reactivity of the rest of the molecule in nuanced ways. Over time, various research teams have pushed these modifications to craft more sophisticated pesticides and high-performance pigments. In my own experience, chasing targeted modifications can sometimes lead you down rabbit holes, but the possibility for innovation keeps teams coming back to this versatile intermediate.
Long catalogs list this compound under a variety of monikers — 2-Chloro-N-phenylacetoacetamide stands out as the most direct synonym, but chemical supply chains sometimes lean on trade names or code numbers. This has caused mix-ups and shipment delays, especially when working with global partners or translating shipment paperwork. Sticking to clear, IUPAC nomenclature and double-checking safety docs saves a ton of trouble down the line.
Working with 2-Chloroacetoacetanilide means taking all the usual precautions given to aromatic amides and chloro-organics. It can irritate eyes and skin, and though it lacks the volatility of more notorious chemicals, airborne dust still represents an inhalation risk in busy plants. Production spaces keep well-marked containers, ventilation, and personal protective equipment as non-negotiables. Over the years, regulatory trends point toward even stricter scrutiny over chloro-organics. Facilities review their practices regularly to ensure compliance with updated local and global rules, especially concerning waste streams and worker exposure. Consistent worker training, periodic air monitoring, and straightforward emergency response plans reflect not just compliance, but commitment to the real people running the show.
Industrial paints and dyestuffs depend heavily on intermediates like 2-Chloroacetoacetanilide. Its role in synthesizing high-performance pigments can’t be overstated — it allows for tougher, brighter, and more consistent colors in everything from auto coatings to plastics. Outside pigments, this compound sits near the root of some active agrochemicals, where transformations on the molecule build to insecticides and fungicides with better crop yields and resistance profiles. R&D chemists often tinker with this intermediate to create samples for new drug candidates or advanced coatings. Any field demanding stable performance and an energetic starting block finds a good partner in this molecule.
Academic and corporate labs continue to study 2-Chloroacetoacetanilide as they search for new reaction pathways or untapped applications. Experienced chemists favor this compound both for its rich chemistry and its relative accessibility. Some efforts focus on streamlining its synthesis to minimize hazardous by-products or environmental impacts. Others tackle the hard challenge of selective functionalization, hoping to open doors in pharmaceuticals or specialty chemicals. It’s easy to dismiss such work as incremental, but years of benchwork often add up to breakthroughs that change what materials are possible at scale. Through my own networking in the community, I’ve seen that sharing results — successes and failures alike — speeds up everyone’s progress and keeps research grounded in lab reality.
Like many industrial intermediates, 2-Chloroacetoacetanilide prompts careful toxicological evaluation. Animal studies and cell assays reveal that acute exposure can cause irritation and, at higher doses, systemic effects. Chronic exposure remains under investigation, but attention focuses on the effects typical of aromatic amides and chlorinated compounds: potential liver or kidney impacts and possible links to genetic changes in vulnerable cell lines. Safety data sheets stress minimizing unnecessary contact and demand respect for disposal protocols. Green chemistry advocates continue to push for alternatives or modified processes that reduce overall chemical hazard. From my experience, trust in a product so often hinges on up-to-date, transparent toxicity data — shortcuts here only invite trouble down the line.
Faced with tightening regulations, plant managers and business leaders look to boost safety and reduce waste from intermediates like 2-Chloroacetoacetanilide. At the same time, demand in coatings, pigments, and crop science shows no sign of fading. Newer synthetic strategies aim to cut down hazardous reagents and energy input, embracing greener routes and smarter, catalytic steps. Researchers eye this molecule as a launchpad for next-gen agrochemicals and novel polymers, betting that small changes to its structure can yield big benefits down the value chain. The best results still come when teams combine good bench chemistry with a clear-eyed look at real-world needs. As anyone who’s worked in an evolving field knows, the future belongs to those who balance performance, safety, and environmental conscience with practical chemistry that gets the job done.
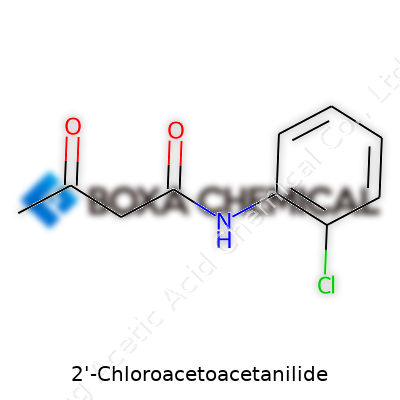
Few people outside a chemistry lab give much thought to how ingredients get their names or formulas. Once a person gets involved in anything related to agrochemicals or pharmaceutical research, though, every letter and number begins to matter. This is especially true for compounds like 2-Chloroacetoacetanilide, which shows up often in the world of herbicide manufacturing. The chemical formula, C10H10ClNO2, isn't just trivia — it’s the thread connecting raw science and real-world solutions.
The logic behind C10H10ClNO2 lies in its structure. There are ten carbon atoms forming the backbone, probably joined into a benzene ring with some branching or substitutions — chemistry textbooks love to show that six-carbon ring as the foundation for endless combinations. This particular molecule brings in one chlorine atom, which isn’t there for decoration. Halogens, like chlorine, get added to organic structures either to tweak a molecule’s reactivity or improve properties like how long it sticks around in the environment. There’s one nitrogen and two oxygens, reminding anyone in the lab that this isn’t just an ordinary hydrocarbon chain. Each element plays a role, influencing things like melting point, toxicity, or how the compound behaves when it hits the soil.
2-Chloroacetoacetanilide pops up mostly in conversations about weed control, thanks to its place in synthesizing certain herbicides. Agriculture runs on chemistry as much as it does on soil. Without compounds like this, crop yields drop and prices shoot up. At the same time, regulators and environmentalists keep a close eye on the use of chlorinated chemicals. One mistake in the formula or a misstep in handling, and the downsides come faster than the benefits.
Anyone who’s worked in a chemical plant knows that accuracy in the formula isn’t academic. Subtle changes in a molecule turn useful products into environmental headaches or health hazards. Stories aren’t hard to find: a switch in position for just one atom can spell the difference between something that breaks down safely in soil and something that lingers in water tables for years. That’s why researchers look beyond formulas and insist on strong safety data. The trick is balancing what works in the field with what’s safe for people and the planet.
Precision in chemical formulas can’t be separated from accountability. If a lab gets it wrong, the impact goes way past lost time. Compounds like 2-Chloroacetoacetanilide may unlock huge value in agriculture or pharmaceuticals, but only if their risks stay managed. Open access to reliable chemical data, tight regulatory oversight, and plenty of continuing education for researchers all help keep trust high and incidents low. Cutting corners may seem like a shortcut, but the cost always comes back around — whether in cleanup bills, lost crops, or risks to workers and communities.
It’s easy to overlook chemicals with long, complicated names, but 2-Chloroacetoacetanilide shows up in places that touch our daily lives more than most people realize. I tend to see conversations about agricultural productivity, environmental impact, and even public health coming back to stories about small molecules like this one.
Crop yields don’t just depend on seeds and rainfall. What happens on the molecular level counts every bit as much. 2-Chloroacetoacetanilide steps in as a key building block for making herbicides. In particular, it’s used as an intermediate in making certain selective weed killers that help safeguard crops like soybeans, cotton, and corn. By starting with this chemical, manufacturers shape substances that target weeds but leave food crops standing tall. The real-world result is more food on tables and fewer chemicals soaking into the ground from broad, indiscriminate spraying.
Colors in our clothing, paper, and even the prints on everyday packaging rarely come from nature anymore. Synthetic dyes create brightness and durability. 2-Chloroacetoacetanilide figures into the story by helping form pigments, especially those that keep their color under sunlight and repeated washing. Whether the shirt you’re wearing is bold red or the book cover on your shelf glows bright blue, there’s a good chance this molecule plays a minor but real part. It allows chemists to create compounds that resist fading, which means better value and less waste.
The journey from raw chemical to medicine can be long, but intermediates like 2-Chloroacetoacetanilide start it off. Its structure helps in synthesizing active pharmaceutical ingredients, particularly where a chloro and acetanilide group provide the right chemistry for further modifications. Bringing down the costs on the chemistry front can open doors for more affordable medicine production down the line.
Every time the chemical industry finds new uses for intermediates, downstream consequences must be considered too. I’ve seen concerns about safe handling, residue management, and the possibility of byproducts entering the environment. 2-Chloroacetoacetanilide holds up as a useful tool, but it reminds us that innovation needs an eye on safety. Professionals handling it use gloves, fume hoods, and good ventilation. Waste must be treated so that nothing leaches into water or soil. Chemistry creates opportunity, but responsibility sits nearby.
Broadly, there’s a crossroads where hope for better yields, longer-lasting colors, and more accessible medicines meets the obligation to reduce harm. Creating targeted herbicides with less runoff means farmers keep both crops and surrounding wild plants healthy. Improved pigment durability translates into less frequent dyeing and fewer environmental impacts from dye factories. Streamlining pharmaceutical synthesis can lower barriers to health.
Still, all these steps demand watchful oversight, open science, and collaboration between producers, regulators, and end users. That’s how good chemistry shapes better futures—by building on past lessons and asking tough questions, every step of the way.
Most folks don’t bump into 2-Chloroacetoacetanilide in their everyday lives, but for those who do, either in research, production, or quality labs, this chemical draws a line in the sand: don’t underestimate its risks. I’ve spent years in a lab where attention to detail mattered more than fancy technical talk. Safety routines surrounding this substance always seemed like overkill until someone made a mistake—a small spill, a missed glove, or a splash in the eye. That’s when the purpose of all those protocols hit home.
In appearance, 2-Chloroacetoacetanilide looks like countless other colorless powders. Its real threat shows up during handling. The substance can irritate the skin, eyes, and respiratory tract. Breathing even a small amount, especially in a poorly ventilated room, leaves your lungs itching and your eyes watering. Absorption through the skin remains a real possibility, meaning those who work with it regularly ought to stay vigilant about exposure, wash hands thoroughly, and avoid eating or drinking anywhere near their workstations.
You won’t catch many chemists handling this compound without gloves, goggles, and a well-fitted lab coat. I’ve learned to reach for a face shield when weighing it out. Nitrile gloves deal with accidental contact, and a sturdy apron helps block splashes. Good, sealed goggles block stray dust and fumes from sneaking into the eyes. For added insurance, some labs bring in a chemical fume hood, not just for peace of mind but to capture dust and fumes before anyone’s exposed. One glassware accident, and you figure out pretty quick why all this matters.
A common mistake crops up at the end of a long shift: rushing the cleanup and storage. 2-Chloroacetoacetanilide calls for a dry, cool spot in a clearly labeled, tightly sealed container. Keep it far from oxidizing agents and far from any moisture. One spill means not just a mess, but possible chemical reactions that make the original compound look mild by comparison. Clear labeling helps new team members, interns, and late-shift co-workers stay sharp, even if they’re tired.
No one wants to come into a lab and sneeze their way through the day because the air’s full of leftovers from yesterday’s job. Working in a ventilated hood prevents buildup in shared spaces. When spills happen, sweeping or vacuuming dry powder into the open air sends it everywhere. Dampening with a bit of wet paper towel, then disposing in a safe waste container, keeps it contained. Don’t treat any chemical like dust at home.
A lot of students and new hires believe chemistry safety lessons are meant for the clumsy or inexperienced. Yet seasoned veterans make mistakes, too. Continuous training, refreshers each year, and clear signage in the lab help drive the point home. Local regulatory bodies update their guidance for a reason. Following that lead isn’t just smart; it keeps people healthy day in, day out.
The best labs run on habits, not fear. Reminding people, checking on each other, and having open conversations after close calls do more for safety than hidden memos or lists buried in a drawer. With 2-Chloroacetoacetanilide, and just about any hazardous material, safety grows from a culture where no shortcut seems worth the risk. Responsibility belongs to everyone, not just the person holding the vial.
Years back, as a grad student in an organic chemistry lab, measuring out the tiniest amounts of different chemicals seemed like a routine step. Yet each measurement carried serious weight—one decimal off, and the experiment failed. The importance of a compound’s molecular weight burned itself into my memory after just one botched synthesis. Even outside the lab, the same principle continues to matter for anyone working in chemistry, pharmacy, or industries producing specialty chemicals.
If you hunt for the molecular weight of 2-Chloroacetoacetanilide, the answer is 211.65 g/mol. This isn’t just a number for the record books. It’s a real tool for anyone mixing batches, scaling up reactions, or figuring out doses. I remember a colleague trying to scale a small lab reaction to a pilot plant—the calculations wouldn’t add up until he realized he was using the wrong atomic weights for the chlorine-containing intermediates. All progress halted till the error got fixed. It’s tedious, but accurate molecular weights are essential to avoid dangerous mistakes and wasted hours in the lab.
The formula comes down to simple addition. 2-Chloroacetoacetanilide’s molecular formula reads as C10H10ClNO2. Breaking it down:
Total: 211.65 grams per mole. This number doesn’t just help with measurements. It supports everything from regulatory documentation to environmental reporting to the patent paperwork for that new dye or pharmaceutical intermediate. Industry experts expect no less, since wrong weights create ripple effects down the entire supply chain. Excesses or shortfalls in a process don’t go unnoticed, and nobody enjoys explaining a costly recall or a failure investigation to the boss or the regulators.
Missteps aren’t just theoretical. Over time I’ve seen analysts mislabel material, labs lose thousands in chemicals from batch errors, even shipments rejected abroad due to wrong specs. One story involved a batch rejected at a port because suppliers listed a molecular weight just a decimal off from documentation. It took weeks and a rush rewrite, delaying everything on the customer’s end. A small thing like a number matters to quality, trust, and safety. To me, the stakes have always been real—accuracy on paper keeps things moving, people safe, and business honest.
Strict attention to numbers means everyone—from researchers to truck drivers delivering the product—relies on solid data for decisions. Successful commercial chemistry runs on repeatability, and trust is built when processes deliver what they promise, every single time. Companies and labs that foster a culture of careful calculation support a safer, cleaner industry. Tools like reliable digital calculators, up-to-date reference books, and transparent protocols make life easier for those on the ground.
Teaching students, I always remind them: the true value of accurate chemistry isn’t limited to formulas in textbooks. It prevents danger, saves money, and keeps businesses and people safe. Speaking from experience, neglecting the details—like molecular weight—never works out well. For 2-Chloroacetoacetanilide or any other molecule, those numbers do the heavy lifting, day in and day out.
Chemicals sometimes hide their dangers behind hard-to-pronounce names. 2-Chloroacetoacetanilide is used by people in labs and factories, especially around dyes and pharmaceuticals. Still, it takes only one careless moment to turn a shelf of powder into a real hazard. From personal time in a chemical manufacturing plant, I’ve seen that nobody forgets the smell of a mishandled container, or the scramble when safety steps get skipped.
Let’s keep it simple: 2-Chloroacetoacetanilide doesn’t like heat. It breaks down faster at higher temperatures. Anything above normal room temperature can speed up spoilage, and the fumes or breakdown products tend not to do anyone any favors for health or purity. I’ve had colleagues find a batch ruined because someone stashed it near a steam pipe. Not only did their work go out the window, they had a messy cleanup with crystal byproducts that really clogged things up.
Sunlight, especially ultra-violet exposure, speeds up decomposition in a lot of chemicals, and this one is no exception. Keeping it in a cool, dark spot works better than risking cracks in glass jars or faded labels. Moisture invites clumping, caking, and surprises during mixing. Even in a controlled lab, I used to line up fresh containers at the start of every batch run—there just isn’t a way to undo the damage from a little water creeping in.
Ordinary plastic often reacts with chemicals. We used thick-walled glass or sturdy, chemically compatible jars with tight-fitting lids. High-density polyethylene sometimes worked, but a risk assessment always came first. I learned to check seals and not to trust recycled bottles, no matter how clean they looked. The cost of a ruined batch always outweighed the price of a good container.
Labeling counted as much as anything else. Handwritten scribbles fade; permanent ink and sticky, chemical-resistant labels saved lots of headaches down the road. I remember one mix-up where someone grabbed the wrong substance because two jars with faded marker ink sat side by side. That led to more than just an awkward staff meeting.
Some teams rely on ventilated cabinets that limit vapor build-up. Good airflow meant less risk to anyone opening a jar. At our site, we logged every removal and return of chemicals, and we held each other accountable for locking everything up at night. Strict inventory checks caught leaking bags before they could cause a problem.
It helped to train everyone, from veterans to new hires, on the real risks tied to poor storage. Mistakes dropped sharply once people understood both the science and the real cost of an accident—not just in dollars, but in wasted time, lost research, and sometimes even burns or breathing problems.
Chemical storage is often treated as an afterthought, yet every worker, product, and manager depends on a little extra care. If one lesson rings true from my years in the field, it’s that safe handling isn’t about paranoid rules—it’s about protecting lives and investments in real, practical ways. No fancy buzzwords, just honest, proven steps—keep it cool, keep it dry, keep it in the right jar, and always put safety ahead of shortcuts.