Chemistry, like most sciences, moves in waves—periods where one discovery sparks a surge of new possibilities. In the case of 2-Chloro-5-chloromethylpyridine, synthetic organic chemists searching for new routes in pesticide and pharmaceutical development flagged this molecule in the late 20th century. Coming off decades where heterocyclic chemistry gained respect for driving innovation beyond dyes and flavors, scientists took to pyridine derivatives for their ability to serve as building blocks. It helped that researchers could produce pyridine cores at scale ever since coal tar research days in the 1800s. By the 1970s and 1980s, laboratories in Europe and North America homed in on this particular chlorinated methylpyridine as a unique intermediate, especially when agrochemical industries pushed for newer, effective molecules in pest management.
2-Chloro-5-chloromethylpyridine isn’t a household name, but its fingerprints show up in products protecting crops or in steps leading to medications. People in chemistry circles recognize it for its role as a synthetic intermediate—a kind of bridge molecule that lets researchers tack on various other functional groups to shape much bigger, often bioactive, compounds. With two reactive sites, it attracts attention for dual functionalization. As farming needs grew more complex, and with growing interest in pest control solutions resistant to older chemistries, 2-Chloro-5-chloromethylpyridine found steady industrial demand. For such a niche compound, its impact stretches further than many realize.
Solid at room temperature, this compound carries a clear pale color and a noticeably sharp odor typical of pyridine derivatives. Its molecular structure—essentially a six-membered ring with nitrogen, both a chlorine atom and a chloromethyl group attached—makes it more reactive than unsubstituted pyridine. The melting point sits comfortably above what you’d expect for a volatile chemical, which helps in handling, but volatility and reactivity demand careful attention. Solubility swings based on the solvent; it manages fair solubility in organic, polar solutions but shies away from water due to those chlorine modifications. Managing the substance requires good judgment and deliberate precautions since the chlorinated structure makes it both useful and potentially hazardous.
Through long experience in laboratory and industrial settings, the main technical benchmarks focus on purity, moisture content, and specific isomeric content. Modern suppliers provide technical sheets with strict thresholds—purity usually not lower than 98 percent for research and synthetic use, often higher when destined for active ingredient production. The proper labeling draws attention to its status as a hazardous material; regulatory bodies such as OSHA and European CLP demand hazard pictograms warning about both toxicity and environmental risks. This isn’t just regulatory red tape. Time in laboratory settings shows that clear, accurate labeling prevents mix-ups and catches the attention of rushed technicians who might forget what sits in an unlabeled bottle. Rigid specification standards go hand-in-hand with responsible handling.
Preparation routes most often use a halogenation approach starting with 2-methyl-5-chloropyridine or a similar precursor. Industry veterans recall days spent tweaking reaction conditions—temperature, solvent, catalysts—to drive selectivity for the right chloromethyl position without over-chlorinating or producing side products. Chlorinating agents combine with methylpyridine under controlled conditions, and through exacting temperature control, the transformation delivers the target molecule. Innovations in greener synthesis have focused on less hazardous chlorinating agents, lower temperatures, and reducing batch waste, responding to both environmental regulations and genuine ethical concerns in modern chemistry.
As a chemical reagent, 2-Chloro-5-chloromethylpyridine doesn’t sit on the shelf for long. Chemists appreciate its dual reactivity—both the pyridine ring and the activated chloromethyl group open up routes to substitutions. Experienced synthetic chemists rely on nucleophilic substitution reactions, often introducing groups like amines or thiols to the methyl position. The chloro attached directly to the ring itself allows coupling or further transformations. My own time in the lab showed that even minor contamination changes the outcome of subsequent steps, teaching respect for tight reaction controls. These functional handles make the molecule valuable in the assembly of agrochemicals and more advanced pharmaceuticals, where reliable, scalable reactions underpin commercial success.
Anyone working in chemical supply or research knows the labyrinth of synonyms that trail every important reagent. 2-Chloro-5-chloromethylpyridine isn’t an exception. Some catalogs list it as “alpha-chloromethyl-2-chloropyridine,” “2-chloro-5-pyridylmethyl chloride,” or similar names. Name confusion leads to ordering delays or worse—wasting grant money and lab time—so double-checking CAS numbers and cross-referencing structure diagrams becomes second nature. In regulatory filings tied to environmental or safety paperwork, these alternative names need mapping to ensure consistency.
Few in industry forget the first experience with chemicals like this—one whiff of pyridine derivatives and you learn fast why handling protocols matter. 2-Chloro-5-chloromethylpyridine poses hazards in several ways: the vapors irritate mucous membranes; direct skin or eye contact triggers chemical burns. Chronic exposure carries even more concerning risks, including potential carcinogenicity, though exact data sometimes trail years behind industrial use. Laboratories and production floors implement forced fume ventilation, gloves, and eye protection. Spill procedures and disposal processes adhere to local environmental rules, reflecting the push in recent years to reduce workplace chemical injuries. This isn’t a mere formality; every safety meeting with real stories drives home the lesson that neglected standards cause accidents and damaged health.
Agrochemical development dominates the market for 2-Chloro-5-chloromethylpyridine. The biggest footprint sits in the production of key insecticides—the neonicotinoid class, for example, often starts from this intermediate or its cousins. These crop protection agents owe their success to efficient, targeted disruption of insect nervous systems, critical for farmers dealing with resistant pests. Outside agriculture, pharmaceutical research values the molecule in synthetic steps for more complex pyridine-based drugs. Chemical manufacturing firms also employ it for specialty ligands and catalysts, tying into broader sectors such as materials science. Its reach spreads quietly, driven by people looking for versatile structures amenable to further modification.
R&D teams focus on several fronts: safer, cleaner synthesis; better downstream chemistry for new crop protection and human medicine; and improved predictive toxicology. University groups and private labs invest time in finding alternative preparation routes, seeking solvents and catalysts with reduced environmental impact. Advancements in computational chemistry improve understanding of how modifications to the pyridine ring and substituents shift reactivity. Collaboration between academic and industrial researchers helps move promising reactions from gram scale to ton scale, where real-world limitations test theoretical breakthroughs. This back-and-forth between fundamental and applied research holds most of the promise for new applications.
Toxicological studies uncover patterns that echo across many industrial intermediates. Some animal tests show the compound can cause organ damage or interfere with central nervous systems at relatively low exposures. Regulatory agencies keep pushing for better chronic exposure data, given the ongoing debate about cumulative effects in humans and environmental persistence. My experience observing regulatory submissions reinforced the need for rigorous, clear reporting, especially as communities near production plants ask tough questions about long-term health effects. Transparent, science-driven risk assessment becomes more valuable as society demands accountability from chemical producers. Industry often drags its feet on new toxicological research, balancing R&D budgets against possible liability, but the safest path forward combines open research with active monitoring.
Looking forward, 2-Chloro-5-chloromethylpyridine will stay relevant as long as the demand for new, effective agrochemicals and pharmaceuticals continues. Environmental and health regulations place more pressure on both producers and users to develop greener, less hazardous processes. There is growing momentum in green chemistry—catalysts that slice down on waste, solvents derived from renewable sources, routes that skip toxic reagents altogether. Digital automation in scale-up processes reduces occupational exposure, a win for industrial safety. Synthetic biology and enzyme catalysis, once limited to simple transformations, begin to show promise for heterocyclic scaffolds like this one. The most compelling changes will likely come from collaborations across disciplines, where environmental science, synthetic organic chemistry, and toxicology integrate to deliver not just better products, but safer and cleaner ways to make them.
Ask anybody who has spent time in a chemical lab about building blocks for agrochemicals and pharmaceuticals, and 2-Chloro-5-chloromethylpyridine often gets a mention. This compound stays in the background for most folks, yet it powers up work that matters in fields from pest control to medicines.
Walking through grain fields in the Midwest, it’s hard to overlook the effort behind keeping crops healthy. The value chain usually starts with compounds like 2-Chloro-5-chloromethylpyridine. One of the main uses connects to the production of neonicotinoid insecticides. These insecticides target insects' nervous systems and help farmers protect staple crops like corn, soybeans, and wheat from pests that would otherwise slash yields. There’s controversy about their impact on pollinators, but from a synthesis perspective, this compound sits at the very first step, giving researchers a reliable starting point to build molecules that help control pests.
For those of us who’ve been caught up in the maze of drug research, versatility matters. 2-Chloro-5-chloromethylpyridine turns into a flexible workhorse for pharmaceutical chemists. Its structure gives scientists a handle for crafting intermediates that end up as antihypertensives, antivirals, and antifungals. The chemistry shows up in practice, not just in theory, as I’ve seen projects jumpstart with this molecule and branch into libraries of drug candidates.
People outside the lab may never hear about intermediates, but anyone who uses pharmaceuticals or enjoys affordable food depends indirectly on these chemical workhorses. Production pipelines rely on compounds like 2-Chloro-5-chloromethylpyridine since they deliver strong yields and predictable reactions during tough steps in multi-layered syntheses. I've watched chemical engineers lean on this molecule, tweaking conditions to deliver purity and quantity for later reactions.
Applications of these chemicals always invite scrutiny. Over the years, several environmental groups and regulators brought up concerns over neonicotinoids due to their link with bee population declines. This has put pressure on chemists and manufacturers to ensure safe handling, search for “greener” alternatives, and think about lifecycle impacts. The central question remains: how to keep reaping the agricultural and medicinal benefits while reducing harm to pollinators, field workers, and communities downwind of manufacturing plants? Smarter process controls, closed systems for handling, and continuous research on less toxic alternatives inch the field toward better outcomes.
After a few years in process development, what stands out to me is the way necessity sparks innovation. The steady demand for high-value crop protection and efficient medicines keeps pushing chemists, toxicologists, and engineers into new territory. Exploring improved catalysts, more selective reactions, and recycling byproducts, labs keep squeezing more value and less waste from each batch. Solutions may not arrive overnight, but the open-ended challenge keeps the field moving forward.
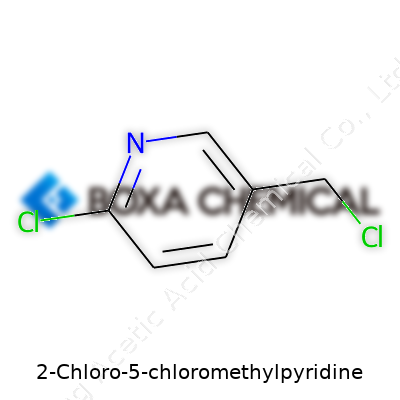
Let’s talk about 2-Chloro-5-chloromethylpyridine, a compound some folks run into in industry or chemical research. Its molecular formula reads as C6H5Cl2N, which tells a story about its structure and chemistry. Each part of this formula points to six carbons, five hydrogens, two chlorines, and one nitrogen gathered around a pyridine ring. People use this compound in making agricultural chemicals, especially as a building block for bigger molecules. Knowing the details here isn’t just for textbook bragging rights—it matters for safety, handling, and ultimately for understanding its behavior in whatever process it’s drafted into.
The molecular weight for 2-Chloro-5-chloromethylpyridine clocks in at 162.02 grams per mole. This number lets chemists measure the exact amount needed in reactions or blend it with other substances without running into surprises. If you’ve ever tried mixing up chemicals in a lab or even just cooking at home, you know exact amounts prevent messes—and big issues in labs, factories, or fields.
Ignoring these molecular basics can cause costly and dangerous mistakes. Someone measuring out a batch for a reaction that doesn’t check the math or trusted a guess instead of the science? That’s asking for trouble, whether it’s an underwhelming product yield or something that goes bang.
2-Chloro-5-chloromethylpyridine isn’t a household name, but it’s got a reputation among folks who spend time with pesticides or specialty chemicals. Safety sheets flag it due to its potential toxicity. Handling starts with the knowledge of its formula and weight, since exposure limits and safety measures stem from those details. It might not act like some notorious chemical villains, but it still calls for gloves, goggles, and respect. Anyone pretending otherwise is risking more than a ruined experiment.
There’s a temptation to gloss over basic properties and skip to practical applications. That approach runs right into problems in real-world settings. A miscalculated dose in a manufacturing process can create byproducts or waste, costing time, money, and sometimes, safety. Agricultural chemists know that safe, effective use of this compound comes from treating the data as crucial, not optional trivia.
Schools and training programs could do more to connect these details beyond dry numbers. We live in a world shaped by chemicals, and the gap between people who understand those details and those who wing it is bigger than it should be. Educational programs should rely on real case studies where misreading something as basic as molecular weight changed outcomes—sometimes with lasting consequences. A strong push for data-driven education lets future scientists, factory workers, and regulators build safer workplaces and products.
Making chemistry more approachable starts with a willingness to see formulas and molecular weights as more than just rote facts. They are starting points for safer workplaces, better products, and fewer accidents in labs and factories alike. The story of 2-Chloro-5-chloromethylpyridine isn’t just about what’s on the label—it’s about what happens when you respect or ignore those details.
Working with chemicals in any lab sparks both curiosity and a healthy touch of caution. 2-Chloro-5-chloromethylpyridine pops up often, especially in pharmaceutical and agrochemical research, because it works as a useful building block for a range of applications. But this compound calls for genuine respect. Its reactivity actually comes through the nose — one whiff can make nearly anyone wince. Breathing the vapors feels unpleasant, and if you’ve worked with strong-smelling agents, you know this usually means real risk.
No one should pour this substance straight from bottle to flask without thinking twice. Long sleeves, solid gloves made from nitrile or neoprene, and fitted goggles beat a quick splash to the skin or eyes. Relying on a well-fitted lab coat goes without saying — after a scare involving a chemical stain that ate through a friend's shirt, I never skipped this step again. Personal experience shows that those who get careless — “I’ll just pipette this quickly” — tend to be the ones sprinting for the eyewash station.
Don’t trust nose or skin as early warning systems. The vapor may hit hard, but harm doesn’t always shout right away. According to the National Center for Biotechnology Information, inhalation and skin contact risks include burns and respiratory irritation, which linger longer than a bad day. There’s no badge of honor in toughing it out. Respirator masks equipped with the right cartridges, plus chemical-resistant aprons, should stay close anytime vapor can waft up unexpectedly.
Fume hoods aren’t just a workplace suggestion, much like turning off the gas stove when leaving home. I’ve learned the hard way: even the best mask won’t make up for circulating sticky vapors in a closed room. Engineers and chemists both rely on local exhaust and negative pressure setups for good reason. Airflow rules still matter in small teaching labs and giant industrial settings alike.
If anyone thinks the cap is tight enough, think again. This compound doesn’t belong near acids, bases, or open shelving by any drain. I keep every reactive agent away from sunlight and heat, double-checking the label before returning it to a clearly marked cabinet. My memory for detail has improved after watching a minor spill corrode the feet on a rolling chair — it wakes a person up to the importance of organized storage.
My intro lab instructor hammered in that chemical spills should never wait for a “good moment.” Absorbent pads, plenty of water, and the right neutralizing agents must be ready before you even break the seal on the container. Nobody ever complained about too much preparation, only too little. Scrubbing spills with gloves instead of paper towels keeps hands clean and nerves steadier, and the Environmental Protection Agency agrees that preventing runoff matters for more than just the lab floor — groundwater and drains don’t deserve a taste of these solvents.
Building a trustworthy lab environment depends on everyone looking out for both themselves and colleagues. The local biosafety authority, the physical safety rep, and the greenest newcomer all play their part. We share stories, cautionary tales, and honest mistakes so others can benefit without making the same errors.
Science moves ahead by asking questions, but some lessons come from old-fashioned caution and a little humility in the face of powerful chemistry. If each person working with 2-Chloro-5-chloromethylpyridine treats it with the seriousness it demands, both workplaces and communities stay safer — and we get to keep learning, mistake by mistake, step by careful step.
2-Chloro-5-chloromethylpyridine holds big value in the world of agrochemical research. Anyone who’s worked with chemicals in the lab or industry knows mistakes in storage can lead to wasted resources, health hazards, and sometimes unexpected headaches. I’ve seen more than a few situations where poor decisions with container choices or ignoring a label resulted in all sorts of problems, especially with reactive or sensitive compounds like this one.
This chemical has a reputation for being reactive. Experience teaches that anything with chloromethyl and pyridine groups demands extra caution. It doesn’t just break down on its own; it teams up with moisture, air, and incompatible materials to cause issues. Let’s be honest: a ruined batch doesn’t just hit your wallet, it puts projects on hold and puts safety at risk. In my early days working with reactive intermediates, I learned fast that turning a blind eye to a leaky cap or stuffy storeroom spells trouble, especially in the summer months when temperatures creep upwards.
Storing this compound means looking beyond the basics. A dry, cool, well-ventilated spot beats a warm, crowded shelf every time. I’ve found that simple steps—like keeping a well-calibrated thermometer close by and checking hygrometers regularly—save more trouble than any quick cleanup ever could. Heat and light act like accelerators, encouraging breakdown and even hazards. Taking that extra step to shield containers from the sun, say with an enclosed cabinet, pays off in the long run.
Proper sealing does more than prevent spills; it keeps out air and moisture. Even if a label claims a container’s tight, I avoid cheap caps and always double-check before putting things away. Once, a rushed intern left a small vial only partially closed, and the humidity drop in quality nearly doubled our costs for that month. Glass beats plastic for chemicals like this, especially with vapor barriers in mind.
Reliable organizations point to the importance of secondary containment, and this matches my own experience. A single spill kit, tucked under the bench and barely used for years, once turned out to be a day saver after a tipped container in a shared fridge. Not only did it shield others from exposure, but it also kept clean-up simple enough to avoid an all-day project. This isn’t about meeting regulations for the sake of it; it’s about respecting the realities of lab safety and keeping hard work safe from setbacks.
More training helps, especially for folks new to handling specialty chemicals. It’s easy to read rules on a screen, but seeing someone else take apart a storage mistake or walk you through why desiccants and secondary containers matter leaves a stronger impression. Investing in laminated guides near storage areas or regular walk-throughs with management sets a tone: quality storage is the foundation of quality science.
Many chemical incidents start small. A forgotten cap, a broken temperature gauge, or a neglected humidity warning can turn a day upside down. By putting practical precautions above shortcuts, and trusting the wisdom of both experienced staff and respected safety sources, anyone working with this compound protects health, budgets, and scientific progress—all at once.
Working in a lab for years changes how you see a chemical shelf. Bottles don’t just hold substances—they send a message about the priorities of the producer. With something as task-specific as 2-Chloro-5-chloromethylpyridine, choices in packaging size and grade signal a lot about the material’s intended journey from warehouse to workspace and eventually down the drain as waste.
When I first started out, I was surprised by how a single compound can come in a dozen bottle shapes and kilogram counts. In practical use, 2-Chloro-5-chloromethylpyridine commonly arrives in amber glass bottles and HDPE drums. You’ll find the smaller packs—ranging from 100 grams up to 500 grams—taking up space in academic and R&D labs. Bulk buyers lean toward larger drums, which can hold anywhere from 5 kilograms on up, sometimes heading to 25 kilograms. This split in packaging volume tracks with the division between those experimenting at a bench and those producing on an industrial scale.
Fine chemical traders and wholesalers keep a close eye on container safety. No one wants their bottle leaking, not because of wasted money, but for the mess and personal risk. Tight-seal lids, secondary containment, and clear hazard labeling provide a layer of trust. Such safeguards go far beyond compliance—they spare accidents in storerooms, fume hoods, and shipping docks.
Purity may sound like a marketing buzzword, but with pyridine derivatives, small differences change everything. In synthesis, every percent of impurity adds cleanup work and can send a project sideways. What most suppliers describe as “analytical grade” or “industrial grade” isn’t just a bureaucratic tick-box; it lines up with real downstream needs. For synthetic work, purity often starts at 97% but can reach as high as 99% for demanding pharmacological or agrochemical applications. A less pure batch might introduce phenolic odors or cloud a reaction, even if it technically fits a looser industrial standard.
Trace metals and byproducts muck up analytics or sabotage catalysts. That hard lesson comes with experience, usually through a spoiled experiment or a batch whose yield falls off a cliff. Every researcher keeps a mental log of which vendors meet their posted specs and which slip. Lab conversations turn into word-of-mouth reviews, giving honest feedback faster than any email from purchasing.
It’s easy to parrot the phrase “quality control,” but anyone who’s run a chromatography shows how tightly things hinge on actual transparency from suppliers. Certificates of analysis don’t always reveal batch-to-batch drift, so labs learn to double-check and sometimes even send samples to third-party testers. The real value comes from suppliers who lay out full batch histories and respond quickly to questions about impurities—no one wants surprises packed inside a barrel.
The current push for better traceability—driven partly by international regulations, partly by grassroots industry demand—means more suppliers now back up their numbers. Regulatory focus on environmental and worker safety points the conversation away from vague claims and toward hard data. Sample retesting, supply chain auditing, and open supplier-client lines all reduce risk for users and producers alike.
Simple improvements make a difference. For example, providing QR codes on bottles gives instant digital access to batch records. Smarter packaging materials—especially antistatic and UV-protective barriers—extend shelf life and reduce waste from spoiled batches. Creating clearer grading systems, not just “technical” or “analytical,” but specifying which impurities matter for what use, would save labs from avoidable headaches.
As someone relying on reliable chemicals day in and out, my main ask isn’t for a glut of options—it’s for honest information and safe, thoughtfully chosen packaging. Every experiment can run smoother and safer if the supply chain keeps user needs—not just profit—at its core.