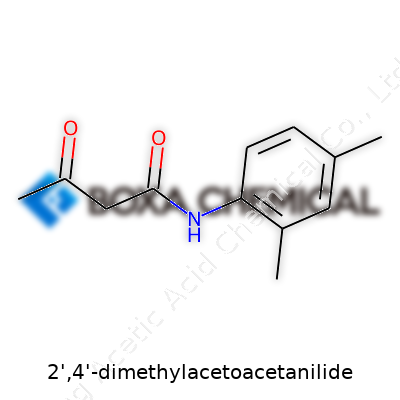
Chemistry textbooks often pack in the intricacies of aromatic ketones, but nothing quite brings home the relevance of 2,4-dimethylacetoacetanilide until you realize how firmly it has planted itself in the story of organic intermediates. Chemical syntheses in the twentieth century made good use of strategies that seemed quite humble by today’s standards, but in the middle of it all, structures like this helped unlock a new class of colorants, pharmaceuticals, and agricultural compounds. Back then, the drive to develop new acetoacetanilides grew out of curiosity as much as necessity. Oil-based industries and growing demand for synthetic dyes and crop protection substances made compounds like this a household name for chemists well before they showed up on patent filings and chemical catalogs. Although history sometimes forgets, much of this molecule’s value comes from work done in a time that cherished careful hands and slow reactions—right before automated chemistry changed the pace of discovery.
After years in labs, one thing I have always appreciated about 2,4-dimethylacetoacetanilide is its predictable profile. Its bright crystalline form tells you right away that this molecule does not shy away from the limelight when it comes to stability under normal conditions. Even in the context of modern analytical techniques, labs can count on reliable melting points, solubility trends, and reactivity under standard reactions. With its distinct methyl groups perched on the 2 and 4 positions of the phenyl ring, it stands as a poster child for structure-function relationships. Each new batch holds a consistency that minimizes frustration and boosts confidence in downstream experiments, making it a steady companion for both seasoned chemists and young hands getting their start at the bench.
It does not take a raft of technical tables to appreciate this compound’s straightforwardness. Years of prepping, drying, and storing 2,4-dimethylacetoacetanilide reassure you that its moderate molecular weight, absence of strong polar functionality, and solid-state form do a good job in day-to-day lab life. The extra methyl groups push its melting point up a notch compared to its parent compounds, making purification work easier. While not hydrophobic enough for some solvent schemes, it dissolves quite comfortably in common organics like ethanol and acetone, which makes for less hassle in reaction setups. Because these properties have been validated countless times, research teams save hours and headaches, focusing their attention where innovation actually happens instead of wrestling with finicky intermediates.
Transparency matters most when dealing with chemicals that may end up in food colors, textiles, or crop sprays. That is why regulatory agencies and responsible manufacturers check for clear, reliable technical information. Each drum or flask should reveal its purity, main impurities, and any residual moisture. Safety labeling needs to keep up with evolving rules, especially as downstream sectors scrutinize supply chains. Personally, I have seen labs scale up production only to face trouble from minor contaminants that never showed up during the research stage. These days, stricter technical specifications and smarter traceability systems prevent headaches before they start, whether for batch-quality assurance or in meeting new environmental rules surfacing worldwide.
One of the pleasures of hands-on organic chemistry lies in seeing a synthesis come to life. 2,4-dimethylacetoacetanilide first takes form through classic acetoacetanilide condensation, where the para and ortho methyl groups on the aniline ring influence not only reactivity but also crystal habits. These days, experienced chemists look for ways to replace old solvent-heavy processes with greener alternatives. In labs like mine, techniques like phase-transfer catalysis or solventless routes slowly take the place of more wasteful methods. I have learned that paying attention to these details isn’t just good for the bottom line; it also prevents unnecessary irritation, contamination, and occupational exposure—all of which matter once you scale up beyond gram quantities.
Chemists tend to admire molecules that lend themselves to creativity, and this one does not disappoint. Thanks to its aceto and anilide framework, a wide menu of transformations opens up. The molecule’s active methylene center acts as an open invitation for alkylation, condensation, and cyclization reactions. Back at university, I once used derivatives of this chemical to make complex heterocycles and pigments that still find commercial use in inks and coatings today. The methyl substituents rarely slow down these reactions—but they also protect against overreactivity, allowing for better selectivity in multi-step syntheses. On the industrial side, this balance between control and versatility means fewer side-products, a point that matters for both efficiency and safety.
The chemical literature overflows with alternative names—some more confusing than helpful. Over the years, I have run into synonyms like 2,4-dimethyl-N-acetoacetanilide, and even some local variants that complicate literature searches and procurement alike. Synonyms reflect both regional naming conventions and evolution in chemical nomenclature. Understanding these aliases matters, not just for students hunting down background reading, but for professionals needing regulatory clarity. Labs and traders benefit from standardizing naming on certificates and packaging, avoiding confusion across borders and academic traditions.
Many of my training hours as a chemist were spent drilling good habits: gloves every time, careful weighing, splash guards when pouring volatile organics. Even though 2,4-dimethylacetoacetanilide does not carry the acute toxicity or volatility of more infamous chemicals, complacency is no friend in the lab. Repeated or careless exposure can build up low-level health risks, including allergies and chronic effects that slip under the radar. Proper local exhaust, good labeling, and hazard communication keep everyone in the loop. Regulations on storage and waste, growing tighter every year, add an external motivation to internal discipline I adopted as law long before enforcement arrived. Well-enforced standards in handling, storage, and disposal reduce not just accidents, but also long-term environmental drift—a persistent concern as more jurisdictions expect transparent reporting from extraction to waste.
Walk into a dye chemistry or pesticide synthesis lab, and odds are you will spot 2,4-dimethylacetoacetanilide on a shelf. Its ancestry ties closely to pigments that color everything from fabrics to printer inks and, in some cases, even food packaging. Its robust functional groups find recurring use where stability under light and moderate temperatures count most—think outdoor paints and papers. My first professional memory of this compound came while troubleshooting pigment formation: batches with it produced cleaner colors with fewer off-shades, a not-so-small win for clients expecting quality every run. In agriculture, subtle tweaks to its skeleton laid the foundation for herbicide ingredients, though increasingly, regulators scrutinize environmental fate and path to breakdown products with a more critical eye. Each one of these practical examples boils down to a simple truth—having a dependable, modifiable intermediate builds confidence in the next step, whether you’re mixing powders for the first time or working up to hundreds of kilos a week.
Research never really stands still, no matter how “solved” a molecule seems to be. In the case of 2,4-dimethylacetoacetanilide, research groups push into unexplored modifications, pairing it with advanced catalysts to build complexity with fewer steps. I see a trend: smaller teams can now tackle transformations that once required massive industrial support. Computational chemistry, together with modern high-throughput screening, guides the search for new biological activities or less hazardous alternatives. The chance to pair old intermediates with new synthetic technologies breathes fresh life into time-tested compounds. Focusing on new crystal habits or molecular architectures, even at the edge of commercial chemistry, paves the way for more sustainable and cost-effective materials, medical probes, or digital printing technologies.
Toxicity sits at the root of every decision in modern chemistry. Decades ago, risk evaluation often relied on trial and error, sometimes with hard lessons for those working unprotected or unaware. Now, risk assessment teams measure not just acute exposures but also chronic impacts on animal and plant systems. Modern studies typically flag the compound for mild hazards—skin sensitization, respiratory issues on dust inhalation, and breakdown by-products that warrant scrutiny. From what I have seen in regulatory meetings and safety audits, companies responding quickly to new findings save both money and reputation. Transparent toxicology, open to peer review, ensures that 2,4-dimethylacetoacetanilide earns its place in chemical inventories and supply chains in a world asking harder questions about sustainability and human health.
No molecule truly marks the end of its journey. In my lab and countless others, 2,4-dimethylacetoacetanilide stands on the edge of major change. New environmental policies, demands for green chemistry, and the push toward biodegradable materials test the relevance of legacy molecules every day. Creative minds now reimagine processes that use less energy, cut solvent waste, and explore hybrid structures meant for a new era of performance materials and safer agricultural solutions. This evolution keeps the field lively—a direct response to both the achievements and shortcomings of the past. Holding to scientific rigor and a willingness to adapt, tomorrow’s chemists build on a foundation that compounds like 2,4-dimethylacetoacetanilide helped pour. Their work points toward a future where old chemistry meets new expectations, and where safety, function, and sustainability push each other forward, step by step.
Most people flipping through catalogs of colorful products or staring at shelves lined with vibrant packaging rarely think about the compounds making those shades possible. 2,4-Dimethylacetoacetanilide, sometimes referenced by the shorter “DMAA,” belongs to this world—one of those behind-the-scenes chemicals pushing the evolution of pigments and dyes further than many notice.
You’ll find DMAA mentioned most often in connection with pigments, especially in the production of azo dyes. Azo dyes rank as the workhorses of synthetic coloring. You find them on the pages of glossy magazines, the plastic wrappers of snack foods, and the fibers of cheap t-shirts. Chemically, DMAA works as a coupling component, helping chemists construct intensely colored molecules that resist fading and blend well into different materials.
As someone who’s spent hours coaxing colors out of raw ingredients in a home studio, I can speak to the frustration of pigments that wash away or fade at the first sign of sunlight. The appeal of strong, stable dyes drives demand for chemicals that promise reliable results. DMAA answers that call. Industrial labs use it to generate pigments with durability, color retention, and cost-effectiveness that natural dyes often lack.
Big textile houses lean on DMAA-derived pigments for more than just color. The clothing industry chases faster production times, sharper colors, and threads that can handle thousands of washes. Compared to old-school natural dyes like indigo, synthetics delivered by DMAA-based chemistry bring practical benefits: faster batch turnaround, better compatibility with synthetic fibers, larger production scales. These don't just save money; they shape global supply chains and the wardrobes filling closets around the world.
Printing ink manufacturers also benefit. Printers running 24/7 crave consistent results—no surprises mid-run, no customer complaints about color mismatch. Pigments born from DMAA do their part, helping deliver results that stay true on everything from cardboard boxes to high-gloss labels. Designers working with many different substrates appreciate the flexibility this brings, knowing that color accuracy won’t get lost in translation.
With these benefits comes responsibility. Any chemical that gets manufactured in bulk and spread across so many industries raises questions. I’ve seen debates in scientific circles and supply chain meetings about the environmental footprint of these ingredients. Factories producing DMAA must invest in safeguards—responsible waste management, air and water treatment, risk mitigation for worker exposure. Regulatory bodies pay attention to persistent organic compounds, and rightly so. People downstream want to know their shirts or packaging aren’t causing unseen harm.
More companies have started publicizing their push toward cleaner processes. Cleaner doesn’t come free, but the demand from consumers and watchdog groups grows every year. There’s genuine promise in green chemistry—finding alternative raw materials, pushing for more biodegradable pigments, dialing in synthetic methods that cut down on toxic byproducts. Chemists in labs and activists on the ground share an odd partnership: both want vibrant colors without a dark environmental cost.
Standing back, 2,4-Dimethylacetoacetanilide looks like another piece in the broad mosaic of chemical industry solutions. It plays a big role in how products catch our eye or endure time and use. But just as important as its effectiveness in making things brighter is the ongoing conversation about responsibility—better oversight, greener options, and a transparent approach to the stories behind the colors in daily life.
The name 2,4-Dimethylacetoacetanilide sits heavy on the tongue. But stripping it down, this molecule draws its roots from the world of dyes and organic synthesis, and its formula — C12H15NO2 — tells a story. My time in the lab taught me that chemical formulas are more than jumble of letters and numbers. They connect to the things we use, the colors around us, and sometimes, to concerns about safety and the environment.
2,4-Dimethylacetoacetanilide’s structure results from a coupling between an acetoacetanilide base and methyl groups placed on the benzene ring, specifically at the 2 and 4 positions. Each part comes with baggage: acetoacetanilides have powered dye-making for decades. Tossing in methyl groups tweaks solubility and how dyes bind to fabric. Nothing in chemistry happens by accident; every atom in that formula pulls its weight in the shop floor, influencing shade, wash-fastness, or how readily a dye slides into waterways.
Anyone who has dyed a shirt knows staying power matters, and for that, stable chemicals like 2,4-dimethylacetoacetanilide have quietly supported the textile industry’s ambitions. Researchers and workers depend on these formulas to produce consistent results batch after batch. I’ve seen firsthand how even small changes in structure ripple into big changes in product performance. One methyl group too many or too few, and you’re back at the drawing board, with wasted time and stained hands.
Constant contact with chemicals brings up worries. Studies trace some acetoacetanilides to possible allergies if not handled right, and the need for personal protective equipment is no joke. Factories that churn out these compounds sometimes spark pollution debates. Textile runoff gone unchecked carries residues that never belonged in rivers in the first place. The formula — those 12 carbons, 15 hydrogens, one nitrogen, two oxygens — looks simple on paper, but its consequences don’t stop at the flask.
I’ve talked to operators and regulators alike. Rules tighten in response to growing health concerns, forcing industries to track every step from handling to disposal. But rules alone do little if the will to enforce and innovate lags behind. Safer alternatives, better practices, and relentless focus on waste treatment can make a difference, not just for those who mix the dyes but for entire communities downstream.
Science keeps evolving, and chemists find ways to reduce toxicity, boost biodegradability, and control exposure. There’s real value in pushing for transparency, whether in publishing full safety data or supporting greener alternatives. I recall the shift toward responsible sourcing in other sectors — the same mindset fits here. Understanding a formula like C12H15NO2 means looking past the page and seeing how small building blocks ripple through economies and ecosystems.
Most folks on the shop floor or in the university lab know the feeling of opening a new bag of chemicals. That slight chemical odor or fine dust floating up isn’t just an inconvenience. Dealing with 2,4-Dimethylacetoacetanilide—used in all sorts of dye and pigment work—underscores what happens when people skip the basics or only half-read the rules.
This compound doesn’t usually make headlines, yet it carries risks. Touching, breathing, or even accidentally ingesting it opens the door to irritating rashes, red eyes, a sore nose, or worse. I remember a coworker getting a nasty skin reaction because he'd handled powders with bare hands, thinking, “It’s just another day.” A trip to the campus clinic and a round of questions later, the message hit home: gloves and goggles aren’t overkill.
Wearing gloves that fit and don’t tear matters more than any warning label. Chemical-resistant gloves—nitrile or neoprene—offer a solid barrier. Some people swap gloves for lab tasks, but that’s just asking for trouble. Once that powder sits on your hands, it clings to pens, doorknobs, or your face, turning a small mistake into a bigger health concern.
Eye protection should become second nature. Splashes or airborne dust catch people off guard. A pair of snug-fitting safety goggles stops a painful incident before it starts. I’ve watched beginners skip this step, only to regret it as soon as a speck lands where it shouldn’t.
Dust doesn’t belong in lungs. Dust masks rated for chemical work, fitted properly, turn a “probably fine” task into one you can finish without a cough. Every inhaled particle increases long-term risk, even if symptoms don’t flare up right away.
Eating or drinking in work areas stores up trouble for later. Residue on hands or desktops moves to sandwiches and coffee cups without anyone noticing. Washing up, especially before meals, helps avoid this invisible route of exposure.
I’ve seen too many workbenches stained with yesterday’s mistakes. Routine cleaning with damp cloths instead of sweeping makes a difference. Dry sweeping stirs up dust you’d rather trap, especially at the end of a shift. Spills need a controlled response; spreading them around just increases risk.
Waste shouldn’t get tossed in ordinary bins. Containers meant for hazardous waste lock up risk for good. It only takes one punctured garbage bag to remind a team what complacency costs.
Good ventilation often gets overlooked, but a steady airflow or a working fume hood pulls trouble out of the picture. Modern labs use fume hoods as background furniture, but the moment that fan splutters, the whole team notices. Fixing broken equipment promptly keeps air quality up and chemicals out of your lungs.
Reading a safety data sheet once doesn't make anyone an expert. Walking through real examples—spills, first aid steps, the right way to store unreleased powder—sticks in your head long after the class. Companies and schools that run hands-on safety drills find their people make fewer mistakes.
Safety depends on habits and expectations. Missing gloves or jammed fume hoods send a message that shortcuts are normal, and everybody pays the price sooner or later. People who speak up about gaps make the workplace safer for everyone—newcomers learn from old hands, and everyone goes home healthy.
Many chemicals don’t capture headlines. They work quietly in labs and on factory floors, without the drama of high explosives or radioactive isotopes. Still, 2,4-Dimethylacetoacetanilide carries its own risks—a white solid, a mouthful of syllables, and a need for respect when stashed away. Some might shrug it off because it doesn’t burn through metal or create billowing clouds of toxic gas on contact with water. Yet, bad storage decisions can turn even mild-mannered chemicals into workplace headaches.
Over the years, I’ve seen crowded chemical cabinets in research labs, where caution faded in the rush to finish experiments before dinner. People left bottles out, lids half-cocked, and powders sitting at eye level. In one case, a co-worker grabbed what looked like a routine compound, because the label smudged from spilled solvent. Uncertainty crept in. Improper storage doesn’t just risk contamination; it makes tracking and tracing impossible during an emergency. A solid like 2,4-Dimethylacetoacetanilide might not prompt alarm bells, but breathing dust from carelessness could irritate skin or lungs, interrupting the quiet flow of work.
Storing this compound really brings basic habits into focus. The best way involves simple, reliable steps: keep it sealed tight in a container of glass or approved plastic, with a clear label that survives occasional splashes and gloved hands. Store on a stable, dry shelf, away from strong acids, oxidizers, or direct sunlight—not next to heating vents or inside cabinets that get steamy. I’ve watched chemicals break down or cake together after a few weeks in a humid room. There’s a reason labs get built with dry, temperature-controlled storage—heat speeds up chemical reactions, and in a stuffy supply closet, that’s asking for trouble, especially over months or years.
Access controls play a role too. I keep certain compounds under lock and key, limiting access to folks who know what they’re handling. Not everyone needs free rein over specialty chemicals, even if they sound harmless. Those misplaced moments—borrowing without asking, moving bottles to odd corners—create confusion and risk later. Tracking who used what and when turns chaos into order. Good recordkeeping helps everyone during audits or investigations.
Nobody wants to slog through more paperwork or inventory checks than needed. But trustworthy systems remove the guesswork from chemical management. Involving the team in annual reviews of storage spaces can uncover hazards before they escalate. Sometimes I ask colleagues to walk through our shelves and hunt for expired stock or cracked jars. That simple act prevented problems from lingering unnoticed. Proper disposal of leftovers, never keeping more than needed, and swapping out aging containers have all made daily work smoother—and safer.
I’ve leaned on reputable sources for guidance, like the National Institute for Occupational Safety and Health and established lab supply companies. They offer best practices not just to tick boxes, but to remind us that protecting people isn’t a chore—it’s a core part of laboratory and warehouse life. Ultimately, storing 2,4-Dimethylacetoacetanilide well isn’t about endless rules. It’s about showing respect for chemicals by building habits now, so safety becomes second nature before a problem arises.
Many folks think of chemistry as something best left to labs, but chemicals like 2,4-Dimethylacetoacetanilide show up much closer to daily life than most realize. Used in pigments, dyes, and coatings, this compound helps keep colors bright on everything from toys to textiles. Over time, I've paid attention to questions that come from both parents and workers: is something in a bright red plastic cup or that new splash of wall paint putting people at risk?
In my years of reading studies and talking to professionals, I keep noticing a gap in how chemical safety gets shared with regular people. Take 2,4-Dimethylacetoacetanilide—scientists know it behaves much like other aromatic amides. Breathing it in, eating it, or soaking it into your skin usually comes from working with pigments at a manufacturing scale. Lab tests don’t show this stuff dissolving easily in water or jumping into the air under normal conditions, so the risk at home tends to stay much lower. Still, factory work changes the picture. Repeated exposure, especially breathing dust or vapors, raises the odds for allergies, skin irritation, or something worse over a lifetime.
Health agencies in Europe and North America don’t rank 2,4-Dimethylacetoacetanilide with their top hazardous chemicals, but they don’t call it harmless, either. Some studies link aromatic amides to changes in blood or liver chemistry and hint at possible long-term risks nobody has ruled out completely. That uncertainty can weigh on people whose work brings them into regular contact with colorants and paints. Protective gloves, goggles, and good ventilation aren’t just red tape—they help protect my friends working in those factories from breathing too much dust or getting itchy, persistent rashes.
Once chemicals slip out of the production cycle, they head towards landfills, wastewater, or just into the air and soil. Most data show 2,4-Dimethylacetoacetanilide breaks down slowly in the environment. That means it can hang around in riverbeds or soil for years, where plants, insects, or fish may pick up trace amounts. This long life cycle raises questions we can’t answer with certainty. Few ecotoxicology studies give clear answers about what this compound does as it builds up in the wild. In places where factories release dye waste, local water sometimes tests high for organic chemicals, raising alarms for both wildlife and people who live downstream.
People deserve to know about chemicals in the air, water, and everyday items—not just when regulators force companies’ hands. It helps to back up those warning labels and safety data sheets with education: what to watch out for, how to store supplies, and even the story behind a compound’s risks. I’ve seen big improvements when companies switch to safer pigments or install scrubbers, and health actually returns for workers who once suffered chronic skin flare-ups. Legislating lower disposal limits and encouraging green chemistry keep factories from pushing the same old risks onto future generations.
Practical changes matter. Good filtration in dye plants, strong training for workers, and more research on how these chemicals move through ecosystems—all work better than scare tactics or sweeping bans. Chemicals like 2,4-Dimethylacetoacetanilide will probably stick around in industry, but awareness and smarter choices go a long way towards balancing color and caution.