The path 2,3,4,5,6-Pentachloropyridine cut through the world of organochlorine chemistry began decades ago, back in a time when the limits of pyridine modification were still getting mapped. In the early 20th century, chemists reached for chlorination as a way to toughen molecular frameworks and explore new uses. By the 1950s, the multichlorinated derivatives had caught the eyes of industry and research labs alike. This particular compound, with its near-full house of chlorines on the pyridine ring, drew notice both for its chemical stubbornness and its potential. Academic records, patent filings, and risk assessment studies all tell stories of how curiosity, commercial interest, and environmental concern kept scientists busy hunting for a balance between benefit and risk.
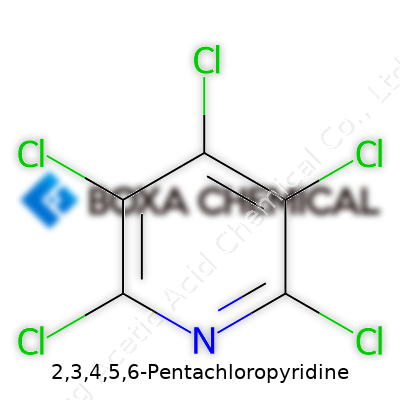
2,3,4,5,6-Pentachloropyridine, as its name spells out, packs a cluster of five chlorine atoms onto a pyridine core. The chlorines punch up the molecule’s chemical resistance, making it less likely to break down in many environments. This stubbornness serves as both an advantage and a challenge. Those looking to exploit its properties see promise in its stability as a starting material for other specialized compounds. On the flip side, this same stability can mean higher persistence in the environment, adding weight to regulatory discussions and green chemistry efforts. Synonyms crowding the registry books—such as Perchloropyridine or hexachloropyridine—reflect a patchy global naming landscape, which can trip up both casual readers and country-to-country shipment records without careful labeling.
Handling 2,3,4,5,6-Pentachloropyridine in the lab brings a set of expectations: it’s a solid crystalline material at room temperature, tinged by a pale beige to yellow hue, and flashes its low volatility in open air. The heavy chlorination drives up its molecular weight, changes its solubility profile, and cranks up its resistance to acids and oxidizers. A compound like this doesn’t mix easily with water, instead preferring the company of organic solvents with similar backbone structures. Its thermal stability allows it to persist at higher temperatures before breaking down, and the smell—the sharp harshness often found with chlorinated aromatics—can hang in the air if not managed with proper ventilation.
Most chemists insist on clear, accurate labeling supported by purity data; no one wants to work with an ambiguous bottle. Analytical verification, whether it be gas chromatography or advanced spectroscopy, comes standard for confirming the exact nature and content of each sample. The fine print on such labels matters. Distinguishing this material from close relatives helps avoid confusion, especially in regulated spaces where small missteps can lead to larger supply chain headaches. Most facilities set their standards high, screening for trace contamination from precursor chemicals and documenting each batch’s journey from raw material to finished goods.
Synthesis usually starts with pyridine, an abundant and versatile heteroaromatic scaffold, exposed to intense chlorination using agents such as chlorine gas, often in the presence of metal catalysts or activated carbon. This isn’t the sort of chemistry most would try outside a well-fitted lab. The process must be tightly controlled to avoid the creation of unwanted polychlorinated byproducts. After the initial reaction, purification often involves crystallization and washing away unreacted materials. Over the years, safer and more selective protocols have entered the literature, prized for their ability to dial down environmental releases and bump up product purity.
Once chemists secure a batch of 2,3,4,5,6-Pentachloropyridine, further reactions open doors. Its high chlorine content lets it act as a launching pad for displacement and cross-coupling reactions. The compound's electron-deficient ring allows for nucleophilic aromatic substitution, which in turn helps build up complex molecules for agrochemical, pharmaceutical, or material science research. Each chlorine atom marks a potential point of attack for those aiming to swap out atoms for something new—broadening the palette for custom molecule design.
People in the chemical industry rely on a patchwork of names, both systematic and trade-oriented, but sloppy handling of synonyms brings risk. There’s little room for miscommunication given the consequences of mishandling a heavily chlorinated product. Regulatory systems and shipping documents increasingly depend on precise registry numbers and standardized names for global commerce and safe use. The scientific community keeps pushing registries and software systems to track renamed compounds, flag duplicates, and steer clear of confusion. Despite best efforts, old habits sometimes sneak in when new employees or international partners use legacy or regional terms.
Direct experience with this compound presses home the importance of rigorous safety standards. The volatility may be low, but dermal and inhalation exposure risk can’t be overlooked. Labs and industrial plants never relax on the need for fume hoods, gloves, and eye protection. Material safety data sheets flag concerns over acute toxicity and longer-term ecological impacts. Beyond the immediate effects on human health, the environmental footprint narrows the margin for error. Waste management protocols include secure storage, contained reactive processes, and systematic handling of residues to prevent uncontrolled release into water or soil. In places subject to regulatory oversight, audits and compliance checks drive the culture of constant vigilance.
Crops and pest management professionals once explored pentachlorinated pyridines as intermediates feeding into complex agrochemicals, especially herbicides and insecticides. The power to resist breakdown, prized for some uses, soon raised concerns over persistence. Today, interests sometimes aim at specialty synthesis—structural motifs in pharmaceutical building blocks, or as intermediates for advanced materials. In my own experience in a chemical research setting, the real test comes down to finding an end-use that justifies the added complexity and environmental weight. With social and regulatory expectations on the rise, the number of routine applications probably shrinks, but niche research projects still ask for small quantities where nothing else will do.
Every time I talk to colleagues in synthesis, the topic of pentachlorinated pyridines sparks debate. Some see them as an irreplaceable building block for new catalysts or high-performance materials. The push goes on to find cleaner routes, safer intermediates, and quicker reaction monitoring. Research teams press for deeper understanding of reactivity patterns, aiming for more selective chlorination and less hazardous waste. Analytical advances let scientists track impurities and reaction intermediates that, decades ago, slipped under the radar. Fresh talent arrives in the field armed with data analysis tools and environmental modeling, sometimes shifting the research priorities toward greener, safer synthetic chemistry.
It’s impossible to ignore the drumbeat of environmental health studies. Persistence in the environment means that any accidental or deliberate release can stick around for years, raising questions about ecosystem and human exposures. Animal studies and cell culture work point to potential risks—liver and kidney toxicity come up often—with data accumulating slowly due to the challenges of tracking and measuring low concentrations across time and space. Regulatory scientists demand convincing proof of breakdown routes before they green-light new plants or manufacturing campaigns. The long memory of environmental contamination events looms large, urging the next generation of chemists to keep safety and stewardship front and center.
Society's appetite for persistent, heavily chlorinated products appears to fade as new green chemistry ideals take hold and more sustainable pathways emerge in industrial design. Academic research pushes for drop-in replacements or modifications that shave off some chlorines, dialing back on environmental permanence. There’s a sense among industry veterans and young investigators alike that the window for large-scale pentachlorinated pyridine use keeps narrowing, unless unforeseen technological leaps unlock safer, truly degradable alternatives. This compound’s future probably lies as a specialty material, studied under controlled conditions, where unique features matter enough to justify careful handling, thoughtful waste management, and transparent reporting.
2,3,4,5,6-Pentachloropyridine doesn’t turn up on average shopping lists, but it plays a background role in chemical manufacturing. Over the years, I’ve seen how niche chemicals like this spark a mix of fear and fascination. Researchers and manufacturers look at it as a gateway for synthesizing bigger, more complex molecules. Its name might trip up the tongue, but the impact stretches into agrochemicals and specialty chemicals.
Most days, people don’t think about what goes into crop protection products or advanced materials. On the factory floor and in research labs, 2,3,4,5,6-Pentachloropyridine pops up as an intermediate—a starting point—mainly for making herbicides and fungicides. Agriculture technology relies on building blocks like this. A molecule with five chlorine atoms hooked to a pyridine ring works well when you need to create tough, stable compounds that stand up in the field.
Science journals outline how this chemical feeds into bigger syntheses. In practice, it lets chemists add chlorinated rings into pesticides. Some of these end-products help guard crops against damage, ensuring stable food supplies. Using such intermediates efficiently, industry can deliver results without reinventing the wheel each season. And for nations where crop yield makes or breaks the budget, streamlined production matters.
Chemicals with heavy halogenation, such as pentachloropyridine, carry edge and risk. Their toxicity and resistance to breakdown often keep them locked inside industrial plants and away from consumer shelves. This isn’t something anyone wants leaking into water tables. I’ve heard colleagues talk about the headaches with safe handling, storage, and disposal. Practice and training set the line between smart use and environmental trouble.
While the push for safer, greener alternatives grows, legacy chemistry can’t just vanish overnight. The European Chemicals Agency and the US EPA watch these substances closely, running risk assessments and urging strict controls. Recent efforts stress developing safer, less persistent intermediates for agrochemical synthesis. Industry leaders and researchers innovate around old molecules, aiming to protect crops without adding burden to soil or water systems.
Some chemicals become notorious; others stay out of the public eye. Pentachloropyridine fits the latter. It doesn’t draw headlines, but its story echoes bigger conversations on chemical safety and sustainable farming. Over the years, watching regulations tighten and markets prioritize “green chemistry,” I see opportunity to shift gears. Investment in better alternatives or redesigning syntheses can help phase out problematic substances.
Knowledge and transparency create trust around chemical manufacturing. Farmers, consumers, and communities deserve clarity on what keeps their systems running. Every molecule, no matter how obscure, carries weight in decisions made both in the lab and on the farm. By pushing for innovation and responsible production, the industry moves toward a safer, cleaner future—even if it means changing the small pieces that build the bigger picture.
Chemicals with long names rarely get attention beyond chemistry labs, but 2,3,4,5,6-pentachloropyridine is lurking in some manufacturing processes that touch the environment and, eventually, people. Behind that complicated name is a substance used in pesticide production and research. Its health and environmental risks push it far beyond academic curiosity.
Drawing from what’s out there, pentachloropyridine doesn’t belong to the family of safe household cleaners. Being highly chlorinated, it shares some traits with notorious chemicals like polychlorinated biphenyls and dioxins. Based on its structure and chemical relatives, there’s good reason to be concerned about both acute and long-term exposure. Reports suggest it can irritate the skin, eyes, and respiratory tract. More troubling is the potential for damage the body can’t shake off easily. Like many chlorinated compounds, it tends to persist—inside us and out in the wider world.
Workers making pesticides, scientists in labs, and communities near production sites land right in the risk zone. The risk does not stop at accidental spills or leaks. Routine disposal or unintended releases can send these molecules downstream into water, soil, and eventually food. These pathways spell out a public health story—one nobody volunteered for, but many could end up living with.
Chlorine-rich chemicals have a reputation for sticking around. Pentachloropyridine tracks a familiar path. It resists breaking down, especially in sediment and soil. Because bacteria and sunlight struggle to dismantle these molecules, the environment holds on to them long after accidental releases. Over time, such persistence means that exposure isn’t just immediate—it’s generational. Wildlife, especially aquatic creatures, face chronic exposure. Toxins move up food chains. Fish provide a direct link between polluted rivers and people’s dinner plates.
The chemical doesn’t work alone. It contributes to a growing load of persistent, bioaccumulative substances in the environment. The best-known problems from these kinds of chemicals include disruption of hormones in animals, decreased reproductive success, and damage to nervous systems. If this sounds familiar, it’s because the world is still reeling from the effects of DDT and PCBs from decades prior.
Some countries have strict rules covering production, transport, and disposal of hazardous chemicals. Yet loopholes stay open for lesser-known substances like pentachloropyridine. Transparency and comprehensive hazard assessments offer the first step. Regulators should demand clear answers from the companies making or using it. Labs and production sites need controls in place—not just for show on safety audits, but to cut down releases and help protect workers.
Communities stand stronger with knowledge. Public health agencies and environmental groups should keep asking hard questions about where these substances end up and in what quantities. Strong monitoring, better waste treatment, and real investment in finding safer alternatives can push industries toward less hazardous paths. Taking these steps seriously beats scrambling to clean up spills, patch together treatment plants, or argue over blame down the road.
Stories about chemicals harming health and ecosystems have played out before. In each case, action came late and costs ran high. With pentachloropyridine, the science may still be catching up, but the pattern looks too familiar to ignore. Pushing for transparency, tougher regulation, and responsible industry practices can spare people and wildlife more harm in the future.
Pyridine rings catch the eye of anyone who’s spent time at a lab bench—an aromatic backbone that’s tough, basic, and used in pharmaceuticals, agrochemicals, and dyes. Now, load it up with chlorine, and it grabs even more attention in the synthetic chemistry world. 2,3,4,5,6-Pentachloropyridine steps in as a striking example: all five available spots on the pyridine ring except for the nitrogen get a chlorine atom. Chemists represent it with the molecular formula C5Cl5N.
So, picture it: a six-membered ring, one nitrogen, and five chlorines slotting into every ring position that can take a substituent. This makes the ring dense with electron-withdrawing groups, and that brings effects that only a handful of other molecules can match. Heavy chlorination isn’t just a curiosity; it defines what the whole molecule does. It's not every day in the lab you find something as chlorinated as this, unless you hang around people working on advanced synthesis or environmental chemistry.
Chlorine changes the game. In college, we’d watch how molecules pick up new habits whenever a new group got added, especially chlorines. For 2,3,4,5,6-pentachloropyridine, those chlorines pull electrons away from the ring, turning it crabby and less likely to share electrons in typical reactions. You can’t just toss it in water or expect it to break down under a heat lamp—these bonds don’t let go easily. The flip side: this sturdiness builds appeal for synthesizing other chemicals that need rugged skeletons, or for crafting novel materials.
On the flip side, stubborn molecules like this don’t leave the environment without a fight. Persistence means risk. Compounds stuffed with chlorines have shown up in waterways and soil, sticking around longer than you’d believe, and sometimes causing harm to wildlife and people. Labs with a green chemistry focus keep their antenna up about what happens to heavily chlorinated chemicals after experiments wrap up.
If you grew up in a time when organochlorines—think DDT or PCBs—made news for bioaccumulating and causing damage, it’s hard not to draw comparisons. There’s a lesson tucked in here: every time a lab builds something like pentachloropyridine, the chemical’s future doesn’t stop at the beaker. There is responsibility involved—what happens during manufacture, use, or in case of accidental spills? Regulators and environmental scientists join in, looking for answers. Some countries draw lines with strict labeling or disposal rules, especially when substances stay in the ground or water for years.
Chemistry classes teach the basics, but the real impact arrives outside textbooks. Where do molecules like 2,3,4,5,6-pentachloropyridine go after they serve their initial purpose? That’s a question that demands honest research and responsible policies. There is ongoing work to build alternatives with less environmental baggage—chemists develop greener routes that trim chlorine use or design molecules that break down faster.
Experience in the lab teaches that clever design can sometimes cut out persistence at the molecular level. It's not about turning away from all useful halogenated compounds, but about thinking ahead—asking if there’s another way before reaching for the five-chlorine solution. That kind of thinking leads to safer processes and less regret down the line.
Safety isn’t a boring checklist—it's what stands between us and a really bad day. Years ago, as a technician in a busy chemical stockroom, I saw firsthand how a little carelessness with hazardous material can blow up into a disaster. Things like 2,3,4,5,6-Pentachloropyridine aren’t just big words on a label; they’re chemicals that demand respect.
Plenty of folks outside the lab haven’t heard of pentachloropyridine. This compound turns up in research and industry, mostly as a building block for heavier-duty chemicals. It's no household item—there’s a reason for that. Each chlorine atom packed onto the ring ramps up the potential toxicity and environmental risk. Breathing in the dust or letting liquid splash on your skin easily means trouble. I’ve watched more than one person ignore the rules, only to wind up needing medical attention.
The rule of thumb: keep pentachloropyridine uncomfortably far from people. Think locked cabinets, clean shelving, and hazard labels you can’t miss from across the room. Dry, cool places ward off unwanted reactions. No: storing near food, drinks, or personal belongings. In my experience, someone always tries to “quickly put something away,” then forgets. So, using a double-check system for storage helps avoid nasty surprises.
Chemicals like this pair especially well with secondary containment. That just means using trays or bins that catch leaks or spills within a cabinet. Some see it as overkill. After cleaning up after a cracked container leaked into a drawer of glassware, I see it as plain common sense.
It’s easy to get lazy with gloves, goggles, and coats. Once, in a rush, I grabbed a flask bare-handed. The stinging reminder stuck with me for weeks. For pentachloropyridine, gloves rated for chemical resistance matter—nitrile or neoprene, not just basic latex. Goggles beat safety glasses every time. Fume hoods keep vapors out of the air you breathe. The National Institute for Occupational Safety and Health has clear evidence: chronic exposure to chlorinated compounds increases cancer risk and damages organs. Using a pipette instead of trying to pour, avoiding any chance of splashing—these little habits lower the odds of a major incident.
People get too comfortable. That’s where mistakes start. Someone forgets to check for cracks in a flask, or rushes the clean-up for lunch. I sat through dozens of safety briefings, but it took seeing a spill go down the drain to really get it. Suddenly, everyone’s scrambling to contain a toxin before it hits the water supply. Old building pipes, flimsy gloves, open flames in the prep area—I’ve seen the whole list of what not to do.
The real solution lives in daily choices. Keep the stuff where only trained folks have access. Lock cabinets. Label everything in plain language. Install a spill kit—absorbent, gloves, disposal bags—right next to the stash, not across the building. Take refresher training every year, not because the rules say so, but because new hazards crop up and old habits need a shake-up. Many labs have started digital checklists to track what goes where and when someone moves a bottle; that extra accountability can change everything.
2,3,4,5,6-Pentachloropyridine is nothing to mess with. With smart storage, serious protective gear, and stubbornly good habits, even the nastiest chemical gets a lot less scary.
Some compounds give off a sense of weight just by hearing their names, and 2,3,4,5,6-Pentachloropyridine is no exception. Picture a pyridine ring, then dress every carbon position with a chlorine atom. The result: a chemical both tough and stubborn, showing little interest in blending with water or fading with time. This molecule crystallizes into a solid, often taking on a pale or yellowish tint. Its density surpasses that of basic organic compounds, a physical “heft” that can’t be ignored in a laboratory setting.
Touching, smelling, or even simply storing such a compound comes with its own considerations. The strong presence of chlorine doesn’t just create weight — it signals reactivity, durability, and sometimes persistence in the environment. It's this relentless chemical grip that makes pentachlorinated compounds the subject of scientific, regulatory, and environmental discussion.
I’ve handled compounds with multiple chlorines in organic chemistry labs, and right away, the challenge becomes avoiding skin contact and inhalation. Chlorine atoms do more than increase toxicity. They ramp up resistance to breakdown, so much that these molecules can last in soils and waterways years beyond their creation date. This inability to degrade means pentachloropyridine, if released, potentially lingers and travels up the food chain.
The chemical world assigns value to such endurance in specific scenarios. Industrially, these types of compounds have played roles in making pesticides, specialty polymers, and dyes requiring fade-resistant colors. On the opposite end, toxicological studies flag high chlorination as a marker for possible bioaccumulation. Labs often require advanced filtration and incineration procedures, since traditional wastewater systems won’t break down these molecules effectively. Safety goggles, gloves, and fume hoods become more than just rules – they’re insurance policies against long-term harm.
Measured melting points often give chemists a direct clue to identity, and pentachloropyridine’s relatively high point reflects the strength and stability of its crystalline form. Its insolubility in water, matched with higher solubility in organic solvents, means that spills behave differently and cleanup calls for specialized protocols. Flammability stays low, but toxic byproducts may release as gases if ignited. Physical robustness doesn’t just test the limits of ordinary laboratory glassware — it also tests the patience of anyone seeking safe disposal or remediation.
Calls to phase out persistent chemicals make sense from an environmental health standpoint, though such measures can’t erase existing contamination overnight. A wiser approach looks to prevention and greener chemistry. Chemists everywhere keep pushing for molecules that deliver the needed durability or color without hanging around for decades unchecked. Researchers design new catalysts to break strong carbon-chlorine bonds, hoping to turn stubborn molecules back into safer building blocks. Better sensors and monitoring tools offer early warnings, helping stop releases before they become problems no neighborhood wants in its backyard.
Understanding pentachloropyridine means facing up to the lasting consequences of our chemistry. Each property — from the way it clings to itself, to its refusal to dissolve in rainwater — speaks volumes about why handling, regulation, and green innovation matter. The push to get it right isn’t just about science; it’s about responsibility and learning from what complex molecules teach us.