Chemistry always tells stories of progress—tales of old labs, persistence, and evolving needs. In the lineage of heterocyclic compounds, 1-(p-Tolyl)-3-methyl-5-pyrazolone shows how certain molecules can carve out distinct chapters in chemical history. Tracing roots back to the golden age of organic synthesis, researchers took a liking to pyrazolones near the turn of the twentieth century, mainly because these hits unlocked new possibilities in color chemistry and pharmaceuticals. The addition of a methyl group and a tolyl ring to pyrazolone’s core didn’t just happen due to idle curiosity. Synthesizing this very compound spoke to a bigger chemical ambition—the need for molecules with sharper, more reliable sensitivity and new scaffolds for drug and dye development. Not one of those classic, overnight breakthroughs, 1-(p-Tolyl)-3-methyl-5-pyrazolone has carried lab notebooks laden with trial and error, each attempt building on the last with tweaks, notes, fresh glassware, and plenty of hope.
Talking about 1-(p-Tolyl)-3-methyl-5-pyrazolone is like looking at a problem-solver among small molecules. Scientists value its rigid ring system and tunable side chains, which make it a cornerstone in analytical reagents. Pulling together chemistry and real-world needs, it’s found in labs as a ligand, an intermediate in dyes, and a skeleton for developing new pharmaceutical candidates. The structure gives it utility for reactions where a stable, robust, and reactive platform proves more useful than flashier designer molecules. Its footprint can be seen in analytical labs where iron content gets measured or where test papers for water quality carry unseen hints of its chemistry. I’ve seen its applications pop up in both early research projects and production processes, a reminder of how seemingly niche compounds often end up touching entire industries.
Getting down to the nitty-gritty, 1-(p-Tolyl)-3-methyl-5-pyrazolone draws attention not for eye-catching color or odor but for solid, practical features. With a stable molecular scaffold, its melting point sits comfortably for most benchwork without drifting into thermal instability. Solubility stays moderate, making it workable with common organic solvents like ethanol or acetone, while water doesn’t readily dissolve it—a trait that can complicate, but also sometimes simplify, workup protocols. This compound doesn’t break down at the drop of a hat, instead standing up to moderate acids and bases. In my experience, that kind of reliability means fewer headaches when scaling up or switching between analytical and synthetic steps.
In lab supply catalogs or on the shelves at research facilities, labels for 1-(p-Tolyl)-3-methyl-5-pyrazolone don’t tend to list endless warnings or obscure hazard codes. Its chemical formula, C11H12N2O, signals a manageable molecular weight, and at first glance, the structure presents a relatively clean handling profile next to more exotic chemicals. Purity specifications often reflect intended use: for critical analytical work or drug research, high-performance liquid chromatography (HPLC) validation sets the standard. Storage recommendations steer clear of high humidity and sunlight—sensibly, since excess moisture can eventually lead to slow-developing degradation. Reading through safety sheets and catalog listings over the years, I’ve always checked whether storage instructions assume students will handle it or if industrial use is more likely, just as a way to gauge the risks.
The real craft with this pyrazolone comes from how it gets made. Lab benches around the world have mixed aromatic aldehydes with methylhydrazine, stirred in some base, and waited for ring closure to do its work. Synthesizing 1-(p-Tolyl)-3-methyl-5-pyrazolone relies on tweaking reaction conditions—temperature, solvent, catalyst—to tip the scales toward yield and purity. Specifics tend to follow a sequence: starting with p-tolualdehyde, reacting it with methylhydrazine hydrate, then cycling the mixture through acidic and basic steps. Sometimes recrystallization in ethanol steps in to clean things up. That experience lines up with what I’ve seen—early runs come out with modest yields, with real progress showing up when reaction times and stoichiometry get dialed in through old-fashioned trial and error. Newer methods swap out harsh reagents, aiming to cut down on waste and simplify isolation, tying into broader goals for green chemistry.
One aspect that keeps 1-(p-Tolyl)-3-methyl-5-pyrazolone on chemists’ radar is how readily it lends itself to further tinkering. Its pyrazolone ring can be modified at several positions through alkylation, acylation, or even coupling reactions, giving rise to a small family of derivatives with varied uses. That flexibility invites researchers to chase activity in pharmaceutical screening or to hunt for sharper color shifts in analytical chemistry. Additions to the tolyl group open up paths for fine-tuning solubility and reactivity. I’ve watched colleagues try out modifications to coax selective metal binding or to stabilize azo dyes for textiles—a real testament to the value of a compound with reliable building blocks and tolerant chemistry.
No one enjoys confusion at the chemical storeroom, and this molecule has picked up more than a few names. I remember seeing “4-Methylphenylmethylpyrazolone” on an old bottle and realizing it traced back to the same compound as “1-(p-Tolyl)-3-methyl-5-pyrazolone.” Some catalogs list it as “p-Tolylmethylpyrazolone” or even refer to code numbers tied to the supplier. These naming quirks run parallel to historical naming conventions, especially from the dye and pharmaceutical literature, where shorthand and systematic labels often coexisted to keep documentation manageable before digital archives ironed out overlaps. Nowadays, cross-referencing CAS numbers helps cut through misunderstandings, but that habit of local naming still pops up in conversation among experienced chemists.
Safety doesn’t just mean the absence of acute hazards—it comes from years of practical observation and following responsible protocols. This compound hasn’t brought headlines for chemical mishaps, but respecting basic rules pays off regardless. Those of us trained in lab safety treat it with gloves and eye protection as a matter of habit and chemistry curriculum. Keeping containers dry and sealed is no shortcut. Dust controls, spill management, and local ventilation have become standard in modern labs, not just to avoid chemical exposure, but to ensure long-term health. Decades of safety culture across the field reinforce that a record of few accidents doesn’t make any fine powder harmless—especially at larger scale or when residues stick around on surfaces. Lab workers benefit most from regular reminders and good design: safe workspaces, proper labeling, and ongoing training mean fewer chance encounters with unexpected problems or forgotten risks.
Plenty of chemical stories start in the lab but actually reach their audience somewhere far beyond, and that goes for this pyrazolone too. Analytical chemists use it to spot trace metals, with test kits relying on its knack for forming intensely colored complexes—a practical tool for environmental monitoring and process control. Its use as an organic intermediate isn’t just academic: dyes and pigments for textiles or inks often depend on reactions starting with this very molecule. In pharmaceutical screening, it provides the kind of backbone that medicinal chemists investigate for new activities, thanks to its manageable size and modifiable structure. From handling real environmental samples to figuring out optimal conditions for azo dye formation, I’ve seen this compound prove itself as both a tool and a stepping-stone toward more specialized products. Its adaptability carries value in both mature and emerging industries.
Authentic progress comes when scientists see a sturdy foundation and try building higher. Ongoing research with 1-(p-Tolyl)-3-methyl-5-pyrazolone has already led to new colorimetric sensors, with improvements in metal ion selectivity and detection thresholds. Collaborative groups are exploring its potential in pharmaceutical leads—pyrazolones have a track record for anti-inflammatory activity, so it’s no surprise to find it tested against various biological targets. Meanwhile, advances in synthesis have cleaned up routes and cut down on solvent waste, inching closer to sustainability goals. Inside these projects, open data and international collaborations have made a visible difference, cutting duplicate work and creating more opportunities for breakthroughs. To stay relevant and attractive for grant funding, scientists keep asking if a simple molecule like this can act as a core for smart sensors, or even get adapted for better solubility in next-generation diagnostic devices.
If you read enough safety sheets, you develop a sense for both what can go wrong and what folks have managed to keep out of the news. 1-(p-Tolyl)-3-methyl-5-pyrazolone hasn’t drawn the kind of regulatory scrutiny or community controversy that befalls more notorious substances, but that doesn’t mean risk isn’t there. Animal studies and in vitro assays dig into how the compound interacts with cells and organs, with eye toward acute toxicity, chronic exposure, and metabolic breakdown. Most results show moderate toxicity, so while it’s not classed alongside industrial poisons, wise handling matters. Research teams reviewing its breakdown products remain alert to any bioactive or persistent fragments, especially if the molecule could leach into water sources or accumulate in biological systems. Transparency about knowledge gaps—especially for newer derivatives—builds long-term safety. That habit helps the field avoid repeating mistakes made with older, less-well-tested compounds.
Looking ahead, 1-(p-Tolyl)-3-methyl-5-pyrazolone isn’t set to fade into obscurity. Analytical chemistry always needs better, more affordable reagents, and this one’s a key player in those colorimetric and spectrometric methods that check water quality or trace metals. The hunt for greener synthesis processes is sparking innovations in how it gets produced, shifting away from legacy methods toward milder conditions and better recycling. In pharma and smart materials, modifications of the core structure promise new patents and maybe even new therapeutic candidates, if safety keeps pace with ambition. As technology stretches analytical demands—smaller samples, trickier matrices—this molecule’s versatility could see it working overtime. Close-eyed research labs, active surveillance of environmental and human health impacts, and open reporting of process improvements will set the tone for how this stalwart compound fits into the next wave of chemical industry advances.
Some chemicals show up in quietly important ways in science, health, and industry. Before I ever stepped into a lab myself, I'd never encountered the mouthful that is 1-(p-Tolyl)-3-methyl-5-pyrazolone. This substance doesn’t get headlines, but it holds a valuable place in pharmaceutical development, especially in the synthesis of pain-relieving and anti-inflammatory drugs. If you’ve read the back of an old box of analgesics, its close relatives might look familiar, since a related compound led to the well-known antipyrine once popular as a fever reducer.
Most folks never see this compound directly; it plays its part in the wings. Medicinal chemists reach for 1-(p-Tolyl)-3-methyl-5-pyrazolone when piecing together complex drug molecules. In my own university labs, we leaned on chemicals like this to introduce key building blocks—imagine a set of Legos that brings pattern and stability to a structure that needs to last under pressure. Analytical and organic chemists value it for forming the skeletons of more targeted drugs.
Over the last century, researchers used this class of compounds to chase after safer, more precise painkillers. For example, derivatives helped pave the way for alternatives when old formulations proved too tough on the stomach or carried risks like kidney issues. The story isn’t just about its historical roots, though; the molecule remains relevant as scientists search for therapies that work better with fewer side effects.
The world’s focus on safer pharmaceuticals keeps substances like 1-(p-Tolyl)-3-methyl-5-pyrazolone in the spotlight of scientific exploration. Pain relief matters to millions who struggle with chronic conditions or acute injury. One lesson I picked up during late nights in the lab—progress in medicine relies on small, often-overlooked components that build toward breakthroughs. The search for less toxic, more effective drugs never ends.
At the same time, handling, producing, and discarding such chemicals responsibly matters, too. Having seen the impact of laboratory waste and community health concerns, I find it crucial to call attention to how standards for production and disposal have to keep pace with innovation. Regulation shaped by research, not just tradition, holds the key. Strong oversight and transparent processes shield both the environment and public health from risks tied to pharmaceutical ingredients.
Solutions to improve safety don’t spring from a single measure. Investment in greener processes allows labs and factories to limit pollution—all while guiding researchers to safer compounds. Support for ongoing education in chemistry, alongside policies that reward innovation, build a system where medical progress and environmental health can reinforce each other.
In the web of scientific discovery and development, substances like 1-(p-Tolyl)-3-methyl-5-pyrazolone remind me that progress grows from layers of small decisions: which route to use, which molecule to build, who gets protected along the way. Respecting that complexity leads to smarter therapies, stronger safeguards, and a deeper sense of responsibility as research turns into daily life.
Anyone working in a chemistry lab or a manufacturing site knows you don’t handle chemicals like 1-(p-Tolyl)-3-methyl-5-pyrazolone with a casual attitude. I’ve seen how seasoned professionals scan labels twice and check protocols before they even crack open a container. The stakes feel personal if you’ve ever watched a coworker make the mistake of skipping gloves, as skin contact or inhalation isn’t something to brush off. This chemical, used in dyes and chemical synthesis, can irritate the skin, eyes, and lungs. You get careless, and you pay a price, sometimes right away, sometimes by the end of your shift.
You find people cutting corners—maybe because they’ve handled similar compounds before or because a supervisor nags less over time. But direct handling of this pyrazolone derivative without gloves, safety goggles, and a lab coat is inviting trouble. I’ve felt the sharp sting of chemicals soaking through a worn-out glove. The difference good nitrile gloves make is clear. Changing them between samples matters. Never touching your face or neck without stripping them off first becomes second nature if you want to stay healthy.
Some people focus only on spills. Reality is, spills are noisy, but airborne dust or vapor creeps up on you. In one instance, I saw a whole team deal with burning throats after a little spill kicked up fine particles while cleaning. If you don’t use a fume hood or at least proper ventilation, you risk breathing in things you can’t see. That risk lingers especially in small prep rooms. I watch for signs of irritation or odd odors, and treat ventilation as non-negotiable.
It’s tempting to stick bottles wherever there’s space. But from my own work, I’ve seen how labeling and sealed containers prevent mix-ups and contamination. Storing this compound along with strong oxidizers or foods in a break room is a recipe for disaster. Segregated storage makes sense—not just for regulations, but for keeping your workmates safe. I remember one winter when a shelf collapsed under the weight of poorly stacked bottles; proper shelving and clear labels saved us hours of cleanup and possible contamination.
Training doesn’t end after a slide presentation on chemical safety. Real learning happens the third or fourth time someone points out your splash goggles are slipping off. You see less experienced staff relax into a routine, and that’s when mistakes tend to happen. In my time, quick reminders and regular refreshers have kept accidents low. No one likes drills, but self-audits and walk-throughs make a real difference. If you’re practicing alone in a space, take breaks to clear your mind—fatigue sneaks up and fogs your judgment.
Accidents still happen. Immediate access to running water and emergency showers is critical. I recall a case when a fast rinse stopped a minor splash turning into something far worse. Knowing where the exits, eyewash stations, and spill kits are, and actually being able to use them, means problems get solved quickly. Relying on memory does not cut it—I’ve walked new colleagues through evacuation routes and spill response steps more often than I can count.
Safety culture comes from peer pressure and good leadership, not just checklists. People should look out for each other. Everyone stays honest when a lab manager rolls up their sleeves and checks every process themselves. Complaints about safety protocols fade when those protocols keep people out of the emergency room. If you want to keep your hands clean and your lungs clear, you respect the compound, follow the steps, and keep each other sharp.
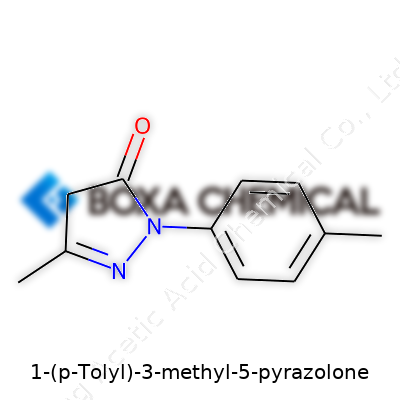
Lots of eyes glance over 1-(p-Tolyl)-3-methyl-5-pyrazolone and see just another chemical name. Beneath the string of syllables hides a structure that turns out to be pretty interesting. This compound, with the formula C11H12N2O, combines two worlds: an energetic tolyl group hooked up with a sturdy pyrazolone ring. I’ve spent time poring over chemical diagrams, and these blended ring systems keep showing up where chemistry meets medicine and color science.
Here’s what stands out to me: the tolyl group—basically a benzene ring with a methyl attached at the para position—gives the molecule a certain magnetism. Chemists know it as “p-tolyl,” and it’s hardly ever just window dressing. The pyrazolone ring, meanwhile, brings its nitrogen double-act and a keto group parked at the five position. That extra methyl nudges things around, impacting shape and reactivity. Across those bonds and rings, the atoms line up in a way that creates all sorts of possibilities for how this molecule can interact with others.
Some might shrug and reach for something more tangible, but it pays to care about what makes up a compound like this. In the late 1800s, chemists stumbled onto pyrazolones and didn’t realize just how useful they’d be. Over time, 1-(p-Tolyl)-3-methyl-5-pyrazolone became a key player in making dyes, especially as an intermediate for azo colorants. Paints, inks, and fibers depend on molecules like these to grab onto vibrant hues and hold onto them through washing, sunlight, and friction. A solid chemical backbone matters if you want colors to last and stay true.
Pharmaceutical research takes a keen interest in this family, too. Pyrazolones show anti-inflammatory and analgesic activity, and the tolyl group can tune a compound's fit with the biological machinery it interacts with. No surprise, then, that a lot of modern drugs link carbon, nitrogen, and oxygen in ways inspired by these classic structures.
I remember poring over reaction schemes and realizing how each group attached to a ring or chain meant a twist in function. Changing a single methyl can move a whole molecule’s behavior—shift its melting point, alter its color, spice up its reactivity. In 1-(p-Tolyl)-3-methyl-5-pyrazolone, the methyl hanging onto the para position gives a boost to how electrons slosh around the tolyl ring, and the pyrazolone core does more than just sit in the middle. Someone working at a dye factory or in a university research group deals with these details every day.
Knowing what’s in a structure like this matters for more than just the lab. Anyone thinking about making safer dyes or better pain medications needs to dig into the relationships between every atom in the chain. We could use this as a jumping-off point—maybe tweak the structure, swap in different groups, and see which changes give better profiles for safety or color stability. Green chemistry circles look for ways to make such compounds with less waste and fewer toxic solvents. There’s a lot to tackle, and chemists need clear, honest conversations about where molecules like 1-(p-Tolyl)-3-methyl-5-pyrazolone take us.
There’s no magic short route around complexity in chemistry. The details of C11H12N2O matter, from the shape of each ring to the electronic push of each substituent. That’s the groundwork that supports progress, both on the bench and in the world beyond the lab.
Some chemicals invite trouble if we treat them like just another jar on the shelf. 1-(p-Tolyl)-3-methyl-5-pyrazolone deserves that kind of respect. I’ve watched more than one promising project at a university slow down simply because someone forgot that even routine storage can shape safety and performance. This compound—found in everything from industrial research to analytics—offers value, but it asks for careful handling.
I remember some folks shrugging off temperature guidelines. A few months later, the sample turned clumpy and useless. This chemical keeps best in a cool, dry spot, not in a place where afternoon heat pounds through a window or where condensation forms along the wall. Extreme temperature swings can nudge chemicals into reacting or degrading—maybe not today, but eventually. So stash it in tightly closed containers, away from steam rooms or heating vents, and keep the lab fridge for something else. I put my trust in shelves tucked in the back, away from the drama of the outdoors.
Nothing ruins a decent chemical faster than water creeping in unnoticed. Humidity makes powders lump together, sometimes triggering slow reactions you might not spot until later. I once opened a bottle of something similar, poured out the clumpy mess, and realized that a month of rainy weather had done its silent work through a loose cap. Tighten the lid. Double-check it. Consider sealing samples in airtight plastic or glass containers with robust gasket lids. Store them somewhere that’s not the first stop for any leaks. Dim, dark cabinets beat bright sunlit shelves. Sunlight drags molecules into breakdown—sometimes creating nasty byproducts nobody ever ordered or wanted to find.
Chemicals left in mystery jars are an accident waiting to happen. Proper labeling, including date of receipt and chemical name, tells everyone what they’re dealing with. I’ve learned the hard way—someone once misread an old, faded label and got a very different reaction than expected. It’s safer to mark things clearly, keep inventories updated, and store new purchases toward the back so older ones don’t sit forgotten until they spoil.
Stashing multiple powders together looks neat, but all it takes is one bit of stray dust in the wrong bottle for a headache down the line. Separate volatile, reactive, or acidic chemicals from something as sensitive as 1-(p-Tolyl)-3-methyl-5-pyrazolone. Even a small contamination, invisible to the eye, can spark trouble or mess with experimental results. Dedicated spatulas, thorough cleaning protocols, and smart container choices go a long way.
Basic storage mistakes often come from taking shortcuts. Ignoring those, for the sake of speed, places people and projects at risk. Institutions keep reminding us—safe storage protects not just samples, but the people working around them. I’ve seen guidelines in action, with small changes making big differences during inspections and audits.
Easy solutions exist: clear labeling, tight seals, dry cabinets, and separating incompatible materials. Awareness and steady habits win out every time. No shortcut beats a safe, thoughtful storage setup for 1-(p-Tolyl)-3-methyl-5-pyrazolone.
Many of the greatest chemical discoveries never started from cutting-edge technology — they sprang from a scientist with a goal and a bench lined with glassware. 1-(p-Tolyl)-3-methyl-5-pyrazolone sits on the shelf as a useful building block, known for its role in pharmaceutical research and, sometimes, for dye intermediates. Anyone with a semester under their belt in organic chemistry knows this molecule comes from pyrazolone chemistry, and the way you make it says a lot about how far chemical synthesis has come.
There’s no escaping the good old Knorr pyrazolone synthesis in undergrad labs. Most routes to 1-(p-Tolyl)-3-methyl-5-pyrazolone start with p-tolylhydrazine and acetoacetic ester. Bring these together in ethanol with some glacial acetic acid, and you’ll see the birth of that yellow pyrazolone ring. After heating, crystals settle into the reaction flask. With a little cooling and filtration, the product shows up in the beaker, just as books promise.
This route brings up a larger issue. Many small-scale academic labs rely on time-tested methods with cheap, off-the-shelf reagents. Purification steps usually involve basic techniques like recrystallization or, if you’re not lucky, a run through a silica gel column. While textbook-perfect, yields can swing more than a volatile stock price, and the process spills some waste — not great for high-volume scale-ups.
Running a reaction overnight and dumping solvent down the drain isn’t popular anymore. Bigger labs and industry chemists have started moving to solvent-minimized, microwave-assisted, and catalytic systems. Some researchers have looked at water-based reactions and even tried ionic liquids, chasing cleaner profiles with fewer byproducts. Avoiding strong acids shaves off some hazards, and microwaves turn long boils into speedy minutes. But high-end setups cost real money, and in practice, not every group has the funding to upgrade.
Every chemist who’s spilled hydrazine on a workbench knows you’re not just dealing with a white powder at the end — you’re handling things that can hurt if you get sloppy. The classic synthesis throws off hydrazine byproducts and uses flammable solvents. Scale that up, and the risks balloon. A move toward aqueous or lower-toxicity methods doesn’t just keep the environment safer; it keeps the person at the bench out of harm.
Green chemistry matters for everyone. I’ve stood at the fume hood, scraping out product and thinking about future regulations that might make waste disposal tougher or more expensive. Fact is, pushing for fewer steps, less waste, and safer starting materials isn’t some academic hobby. It saves headaches, fees, and sometimes lives over the long haul.
Someone picking up this synthesis for their first research project probably won’t invent the next Nobel Prize reaction. Still, sharing better, safer protocols gives students and researchers a hand up. Group leaders can push for collaborative research on more sustainable conditions. Journals and conferences need to highlight green alternatives and publish honest metrics on waste and yield, not just glamour shots of pure product.
Things may not change overnight, but every small improvement in how we make molecules like 1-(p-Tolyl)-3-methyl-5-pyrazolone helps move chemistry in a cleaner direction. Sometimes, that means dusting off old glassware and rethinking the basics with a sharper eye for people and the planet.