It’s easy to miss the inside story on a compound like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone until digging a little deeper. Pyrazolones, as a class, cropped up in European scientific circles over a century ago. Researchers in the late 1800s zeroed in on these nitrogen-containing rings because they opened doors both in medicine and dye chemistry. Moving into the twentieth century, someone tweaked the core, adding a sulfonic acid group. Suddenly, the simple pyrazolone gained water solubility and an edge for industrial synthesis. Over the decades, chemists used it more and more in analytical chemistry—especially in colorimetric assays—and it quietly became a staple for labs that need fine-tuned detection methods. The journey from bench curiosity to practical ingredient speaks to the value of modest, incremental chemical innovation rather than flashy breakthroughs.
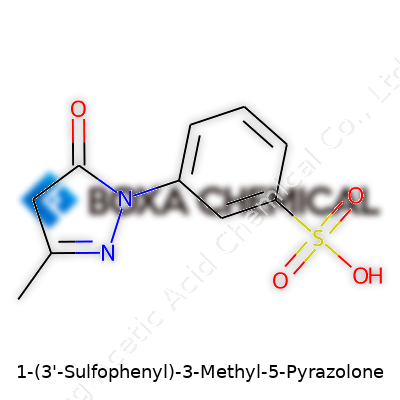
Looking at 1-(3-sulfophenyl)-3-methyl-5-pyrazolone, you won’t mistake it for a substance common in household cabinets. The molecule gives off a yellowish tinge, commonly crystallizing as a powder that dissolves easily in water because of its sulfonic acid group. Chemists note a sharp melting point, which makes it handy for purification by recrystallization. You also see it standing up to air and light fairly well, meaning shelf-life extends practically, without specialized storage protocols. The chemical structure looks simple at first glance—a five-membered pyrazolone ring attached to a methyl group on one end and a 3-sulfophenyl on the other—but the way that sulfonic acid swings the molecule’s properties has fueled its adoption.
Every lab that works with specialty pyrazolones expects clear labeling. Detailed product documentation covers impurities, residual solvents, and storage advice. Industry standards emphasize a high purity level, often 98 percent or above, as even minor contaminants can skew analytical results. Certificates usually detail the batch’s melting point, moisture content, and identification using IR or NMR spectroscopy, reflecting a culture of transparency that experienced chemists have come to trust. This is more than box-checking—it’s mutual protection for lab users and their research integrity.
Preparation methods have grown comfortable with the paths laid out by older chemical literature. Synthesis starts from 3-sulfobenzaldehyde and 3-methyl-1-phenyl-2-pyrazolin-5-one, under typical condensation conditions. Careful temperature control, slow addition, and pH monitoring reduce by-products and boost yields—a nod to decades of trial and error. Recrystallization in water or a water–alcohol mix rounds out the process. This may sound routine, but in practice, production at any real scale means thinking hard about solvent recycling, minimizing waste, and keeping costs down. I’ve worked in a lab where solvent smells lingered for days—every extra step in purification and every kilo of solvent saved means more than just staying on budget: it’s about working smarter and respecting the environment.
Once in a beaker, 1-(3-sulfophenyl)-3-methyl-5-pyrazolone shows it doesn’t just sit idle. Its reactive sites invite further chemistry: the pyrazolone ring may undergo alkylation, acylation, or azo coupling, while the sulfonic acid enables salt formation with alkali metals or bases. Analytical chemists have leaned hard on this flexibility. They build sensors, tweak the molecule’s sensitivity, and tailor analytical standards for water analysis, metal ion detection, or even pharmaceutical quality assurance. The drive for modifications comes directly from user experience—if a molecule gives better color response or less interference from water impurities, chemists run with it.
Synonyms for this compound list out over time and across borders. Some call it Methyl Orange Pyrazolone, others recognize the extended versions describing its functional groups. For procurement, finding the synonym matching your regional catalog avoids days lost in translation. The variety of names reflects broad use, but also a patchwork of documentation—one of those real-world headaches any seasoned scientist learns to navigate.
Users in any chemical setting know that chemical safety isn't theoretical. Pyrazolones rarely top the toxicology charts, but the sulfonic acid group can still cause mild skin or eye irritation. Wearing gloves, goggles, and lab coats isn’t up for debate; neither is mindful waste disposal. European and North American safety sheets spell out handling, storage, and spill clean-up with unambiguous clarity. I remember a minor splash during a rushed run—seconds spent rinsing and reporting kept it a non-issue, but the experience sticks as a reminder that familiarity can breed inattention. Real safety arises from habits built in training and reinforced every day.
This compound shapes up as more than a chemical oddity. Analysts use it in water-testing kits for quantifying trace metals like iron—a role bolstered by the compound's vibrant color changes when binding ions. Some industrial labs harness it for spectrophotometric calibration, while specialty ink or dye manufacturers note its ability to interact predictably with other colorants. Its robust water solubility and well-mapped reaction profile have made it a toolkit staple for soil testing, trace analysis, and standardization processes. Real-world problems—ensuring accurate measurements in municipal water, or flagging heavy metals in food—find everyday solutions in chemistry like this, making the case for supporting niche compound availability.
Research into 1-(3-sulfophenyl)-3-methyl-5-pyrazolone spins out in more directions than seem obvious at first glance. Projects focus on signal optimization in colorimetric tests, extending shelf life in challenging climates, or sharpening selectivity for tougher samples. Some scientists experiment with embedding it in matrices or creating polymer supports for easier industrial-scale filtration. Academic labs still publish improvements on the synthesis route, cutting out steps or boosting atom economy. Open-access chemical databases promote shared findings, and crowdsourcing data on efficacy or trace contaminants creates shortcuts for newcomers. The culture of chemical R&D around these so-called legacy compounds always builds from what came before, but never settles into complacency.
Toxicity studies, albeit scattered, show low acute toxicity but urge caution around chronic exposure and cumulative environmental release. Wastewater treatment plants must strip out aromatic organics, and some of these molecules, including modified pyrazolones, resist standard biological breakdown. Regulatory guidance pushes for containment and best-practices disposal. The past decade’s focus on green chemistry brings persistent chemicals under tougher review; solvents, process water, and final residues grab the attention that decades ago went elsewhere. Responsible scientists—at all stages—know that environmental duty links directly with product stewardship.
The future of 1-(3-sulfophenyl)-3-methyl-5-pyrazolone doesn’t rest on a single breakthrough. Incremental progress—like more robust syntheses or refined detection limits—keeps its use relevant. The rise of portable and field-ready chemical sensors points to new markets outside the lab, especially in developing regions where cost and reliability trump advanced instrumentation. Renewed attention from regulatory science and environmental chemistry stands ready to push greener reagents and safer handling protocols, ensuring the compound stays in the toolkit without tipping regulatory balance. What matters here is that tools supporting clear analysis and data integrity always find a way to persist, even as flashier substances and headlines come and go.
Trying to explain why 1-(3-sulfophenyl)-3-methyl-5-pyrazolone matters can feel daunting until you start seeing where it shows up. Known to most in science as a clever chemical, this compound hides behind the curtain in labs around the globe. It mainly shares the backstage with diagnostic testing, helping doctors figure out what’s happening inside the body. It's not part of a headline yet plays a strong supporting role in medicine every single day.
Anyone who knows someone with diabetes has probably heard about blood tests monitoring glucose and enzymes. Labs don’t rely only on fancy machines; simple chemistry steps up in many cases. Here is where 1-(3-sulfophenyl)-3-methyl-5-pyrazolone fits in. As a reagent, it reacts with compounds in blood or urine to help scientists and technicians detect even tiny traces of substances. Good test results depend on accuracy and reliability, both of which hinge on the tools being used. This chemical tool gives a clear, measurable result so doctors can spot health changes early on.
Though science gets cleaner every year, all chemicals come with their own problems. Labs produce waste and spend money dealing with disposal. Some chemical residues can build up in water and soil when managed poorly, causing headaches for environmental regulators and local communities. My own city faced shelter advisories near water processing facilities that traced back to neglected waste from industrial labs. While the compound itself doesn’t tend to make news for violence or toxicity like some others, every widely used synthetic material brings stewardship issues—especially if scale grows over time.
Not everyone grows up knowing their way around a chemistry lab. People trust that their health data gets measured with precision, and 1-(3-sulfophenyl)-3-methyl-5-pyrazolone helps keep that trust. Adding more oversight on handling and disposal always counts for something, especially in countries where laboratories are popping up fast and supply chains stretch further every year. Education could help, too. Even quick training sessions on storage and waste can keep labs on the right side of safety while respecting the environment.
Technology keeps getting sharper, and so do choices around chemicals. Green chemistry efforts focus on inventing smarter reagents or finding ways to recycle results into new products. Some labs close the loop by reclaiming chemicals from used tests, reducing their footprint without giving up the accuracy science and medicine need. Progress here blends lab innovation with stronger regulation and simple awareness. For those with family, friends, or neighbors working in diagnostics, their job depends on chemicals like this—and on society expecting smart stewardship. It starts with a single compound, but the mindset carries far beyond the bench.
Some people shrug off chemical safety as if fumbling with glassware plus a lab coat solves everything. Reality hits different in a real world lab—especially when you’re staring at an open container of a substance like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone. I remember the sharp, almost metallic odor stepping into the room. Anyone who’s spent a good chunk of time in a research environment knows skin contact, inhalation, or a splash in the eye can ruin an afternoon or worse.
This isn’t just about not getting burned or itchy. Many folks don’t realize that some compounds cause delayed reactions or bioaccumulate after repeated exposure. 1-(3-sulfophenyl)-3-methyl-5-pyrazolone is used in chemistry settings and as a reagent, showing up in colorimetric analysis or more specialized benches. Protective gear doesn’t only help in the obvious splash moments—it stops the small exposures that add up.
I get the temptation to skip gloves or dial back on masks, especially when the room heats up and everyone is on a tight deadline. But mistakes pile up whenever shortcuts creep in. Nitrile gloves give a decent barrier; lab coats go a long way in stopping splashes on your skin and clothes. If there’s even a chance of dust or vapor, a well-fitting lab respirator does more than a flimsy surgical mask.
Fume hoods exist for a reason—and not just for the stuff with a skull and crossbones. Powdered chemicals easily form dust clouds, sometimes invisible, which settle everywhere. Researchers far smarter than me have ended up coughing their way through the afternoon because they thought “just a quick measurement” would be fine in the open air. Never worth it.
Chemical safety isn’t just about one day or one container. Routine means something. Before handling 1-(3-sulfophenyl)-3-methyl-5-pyrazolone, I always double check for leaks, check storage labels, and run through the MSDS. Some say it’s overkill, but the best research teams I’ve worked with never leave a thing to chance.
After use, waste disposal matters as much as anything else. Dumping old reagent down the drain doesn’t just break local waste codes—it puts the next person in danger. Properly labeled chemical waste containers and regularly scheduled pickup prevent ugly surprises.
Too many labs still treat safety as an afterthought or a formality for audits. That attitude sells everyone short. I’ve seen junior researchers get sidelined by chemical burns, and watched labs lose critical time after an avoidable exposure incident. Investing in regular training keeps everyone sharp. Those repetitive drills? They stick when a genuine accident happens.
Regulations exist for a reason. Anyone who’s dealt with even a minor incident knows paperwork and investigations slow progress more than spending an extra minute to wear goggles or wipe down workbenches. Cultivate an environment where people speak up when something feels off.
The best labs don’t just make PPE and ventilation a rule; they make safety part of everyday habits. Open communication, shared responsibility, and hands-on supervision keep risk at bay. It’s not about paranoia; it’s about respect—for yourself, your team, your science. I’ve worked in spaces where safety culture actually drives better science, builds mentorship, and helps younger researchers get their feet wet without stepping into danger.
Taking 1-(3-sulfophenyl)-3-methyl-5-pyrazolone, or any lab chemical, seriously can transform work from risky business to sustainable progress. No experiment is worth trading your health. Science needs minds—not avoidable injuries.
Nobody walks into a laboratory raving about 1-(3-sulfophenyl)-3-methyl-5-pyrazolone at dinner parties, but beneath its clunky name, this molecule shapes more corners of science than you’d expect. Chemists and biochemists often knowing it from dyes or pharmaceutical intermediates, see it as a workhorse, not a celebrity. Still, its structure does a lot of heavy lifting.
Breaking it down, the heart of this compound is a pyrazolone ring. That’s a five-membered ring—two nitrogens, three carbons—fused with some interesting tricks: a methyl group at position three and a phenyl ring at position one. The phenyl ring itself is “sulfonated” at the third position, meaning a sulfonic acid group (-SO3H) is hanging off it. Together it forms the structure: C10H10N2O4S.
The story of pyrazolones goes back to the 19th century, when chemists hunting for fever reducers and dyes stumbled on them. The 3-methyl-5-pyrazolone skeleton turns up in pharmaceuticals and imaging agents. With that sulfo group, this specific molecule dissolves well in water—a trait that can speed up processing in both industrial and clinical labs.
That solubility puts 1-(3-sulfophenyl)-3-methyl-5-pyrazolone in the toolkit for making azo dyes and certain analytical reagents. It crops up in methods for measuring things like bilirubin and phenol. I’ve seen biochemistry students groan when they realize these molecules aren’t just theoretical puzzles—they’re practical components in some of the blood panels run every day at hospitals.
The practical side of this chemistry lands on two main stages: industrial colorants and clinical chemistry. For doctors running liver function tests, 1-(3-sulfophenyl)-3-methyl-5-pyrazolone sits at the center of the Jendrassik–Grof method for measuring bilirubin. My experience with lab diagnostics has taught me that reliability and specificity mean everything in clinical settings. False positives or imprecise results can muddy the decisions doctors make, so stable, soluble reagents like this one play a quiet but vital role.
On the industrial front, the molecule’s zwitterionic nature helps it act as a dye intermediate—improving color brilliance and water resistance in textiles or inks. The world’s appetite for sharp, long-lasting colors in everything from clothes to packaging owes a debt to small, highly specialized organic compounds. The environmental side of dye chemistry can get dicey, though, since sulfonated organics persist in wastewater and call for well-designed treatment processes.
Science circles keep pushing for safer dye pathways and greener synthesis, urging a shift away from pollutants that linger. Techniques for improved water treatment and cleaner chemical substitutions make sense for both regulatory reasons and community health. Chemists in both industrial and academic spaces need to think about not just color performance, but also biodegradation and lifecycle.
For anyone curious about chemistry in the wild, this molecule reminds us how scientific advances ripple out into everyday life. Compounds like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone aren’t the showiest, but their impact calls for both old-fashioned respect and modern accountability. Better practices in lab and factory settings can keep the benefits while minimizing the fallout—balancing performance and responsibility where science meets society.
Storing chemicals like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone sometimes gets treated like an afterthought. People toss them on a random shelf, slap on a label, and move on. The real story unfolds later, when moisture sneaks in, or sunlight heats things up, or worse, vapors collect. I've learned this lesson in labs where attention to detail makes the difference between safe storage and expensive accidents.
This compound won't handle humidity or direct sunlight well. Deterioration sneaks up when storage rooms get muggy or a heat wave pushes temperatures out of the safe range. In my past work with specialty chemicals, stubborn yellowing or even caked powders pointed straight at bad storage. A dry, cool place offers the best hope—preferably, shelving in a dedicated chemical cabinet with air conditioning or, for some labs, at least a stable basement environment. It’s not about high-tech solutions; it’s about discipline.
I remember digging out an old bottle once, crusted around the rim. Stuff like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone begs for careful sealing, with screw caps that actually fit and show no cracks. Containers made of glass or certain plastics work best, since these resist chemical reactions far better than others. Labels need to stay visible and accurate; faded writing or missing hazard signs can get people hurt or trigger confusion during inspections.
Crowding chemicals together turns a safe shelf into a roulette. This compound's sulfonate group means it's water-soluble, so cross contamination with water-reactive chemicals or organics could cause big problems. Segregate storage: acids and bases don’t hang out, and oxidizers get their own spot. That lesson came home once during a shelf-cleaning project, when we found a sticky mess from spilled solvents mixed with a stray powder.
Sure, local rules can seem like legalese, but they’re built from hard-earned safety lessons. Material Safety Data Sheets (MSDS) spell out what works—don’t improvise. Wear gloves and safety glasses just handling the bottle for inventory or transfer. Engineers and lab techs I know always keep PPE close, mostly because one slip can set projects back weeks or trigger a shutdown.
People chase fancy monitoring systems, but routine beats technology every time for basic storage. Check containers each month for leaks or brittleness. Sharpen up documentation. Teach newcomers not only what to do, but why. Lab culture shifts when everyone sees safe storage less as red tape and more as a foundation for good science.
Many younger staff never see the nasty side effects of careless storage because, frankly, folks before them did things right. Clean habits, regular checks, and a cool, dry space protect not just employees but the research itself. Problems avoided never make headlines—but that’s the point.
Anyone digging into chemical safety comes across names like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone. This compound shows up often in laboratories and dye industries, and some may know it as a reagent in colorimetric tests for metal ions. It’s not a household name, yet it sits quietly behind more recognizable results: clean laboratory protocols, vivid pigments, and tidy research papers.
Before picking up a bottle with a label reading this mouthful, it’s smart to ask hard questions about its potential health and environmental risks. Few non-specialists ever talk about such chemicals unless there’s a crisis or an incident at work. Yet leaving these conversations to regulatory bodies doesn’t do anyone much good.
Extensive reviews of published data and chemical safety authorities reveal limited but notable hazards linked with 1-(3-sulfophenyl)-3-methyl-5-pyrazolone. Acute toxicity in animal studies indicates moderate oral danger, which means a lab worker accidentally swallowing even a small amount faces real health problems. General symptoms reported in similar compounds include gastrointestinal irritation, headaches, dizziness, and in rare cases more serious systemic effects. The compound can trigger eye and skin irritation with direct contact—no surprise for anyone who has worked with organic reagents before.
No credible evidence links this particular molecule to chronic health issues or strong carcinogenic properties, but this doesn’t give it a clean bill of health. Less data doesn’t mean less danger, it just points to fewer studies. Responsible users never let gaps in research lull them into a false sense of security. The European Chemicals Agency and other stewardship groups generally urge the same wisdom: treat all new or poorly understood chemicals with caution until proven otherwise.
Spills present their own headache. Disposing of this compound through the drain leads to questions about breakdown in wastewater systems and the fate of breakdown products in rivers or soil. The compound is sulfonated and water-soluble, so it likely travels easily through water systems. Luckily, strong environmental regulations in much of Europe and North America force businesses and laboratories to filter, neutralize, and document chemical waste rather than dump it.
Workers also face exposure through accidental inhalation of fine dust or mist. Even a light breeze across an open container can stir up trouble. Personal protective equipment comes into play: gloves, goggles, and proper lab coats. None of these protections function unless workers get consistent training and the right materials to clean up spills or contain dust. The safest lab I worked in posted step-by-step emergency instructions at every sink and always made sure we had spill kits within arm’s reach.
Discussion around chemical safety often hits a wall as budgets and workloads balloon. As someone who cut their teeth in chemistry teaching labs, I saw how shortcuts crept in when supervisors faced tight timelines and scarce resources. At the same time, students and junior staff sometimes handled reagents with casual carelessness—until they saw a mishap or learned about near misses in a meeting after hours.
Peer learning goes a long way toward preventing accidents. Sharing stories can make safety real—much more than any set of printed instructions or boilerplate warnings. In my experience, regular training refreshers, combined with an open-door policy for reporting hazards, create a culture where people speak up before something turns costly or tragic.
In the end, safety for chemicals like 1-(3-sulfophenyl)-3-methyl-5-pyrazolone never comes down to just reading a data sheet. It grows from constant vigilance, honest communication, and enforced standards backed up with real resources. Everyone in the chain, from managers to interns, shoulders a piece of that responsibility.